Bond length of H H is 0.64 and the bind length of F2 is 1.2. Electronegativities of H and F respectively are 2.1 and 4.1.What is the bond length of HF? 1)0.64 2)0.92 3)0.82 4)0.62
Bond length of H H is 0.64 and the bind length of F2 is 1.2. Electronegativities of H and F respectively are 2.1 and 4.1.What is the bond length of HF? 1)0.64 2)0.92 3)0.82 4)0.62
Bond length of H-H is 0-64 and the bind length of F2 is 1-2- Electronegativities of H and F respectively are 2-1 and 4-1-What is the bond length of HF- 1-0-64 2-0-92 3-0-82 4-0-62
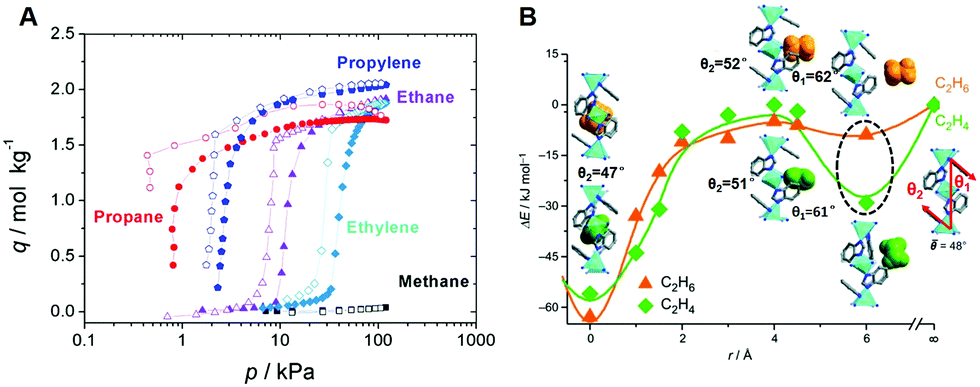
Energy-efficient separation alternatives: metal–organic frameworks and membranes for hydrocarbon separation - Chemical Society Reviews (RSC Publishing) DOI:10.1039/C9CS00756C
9.10: Bond Energies and Bond Lengths - Chemistry LibreTexts
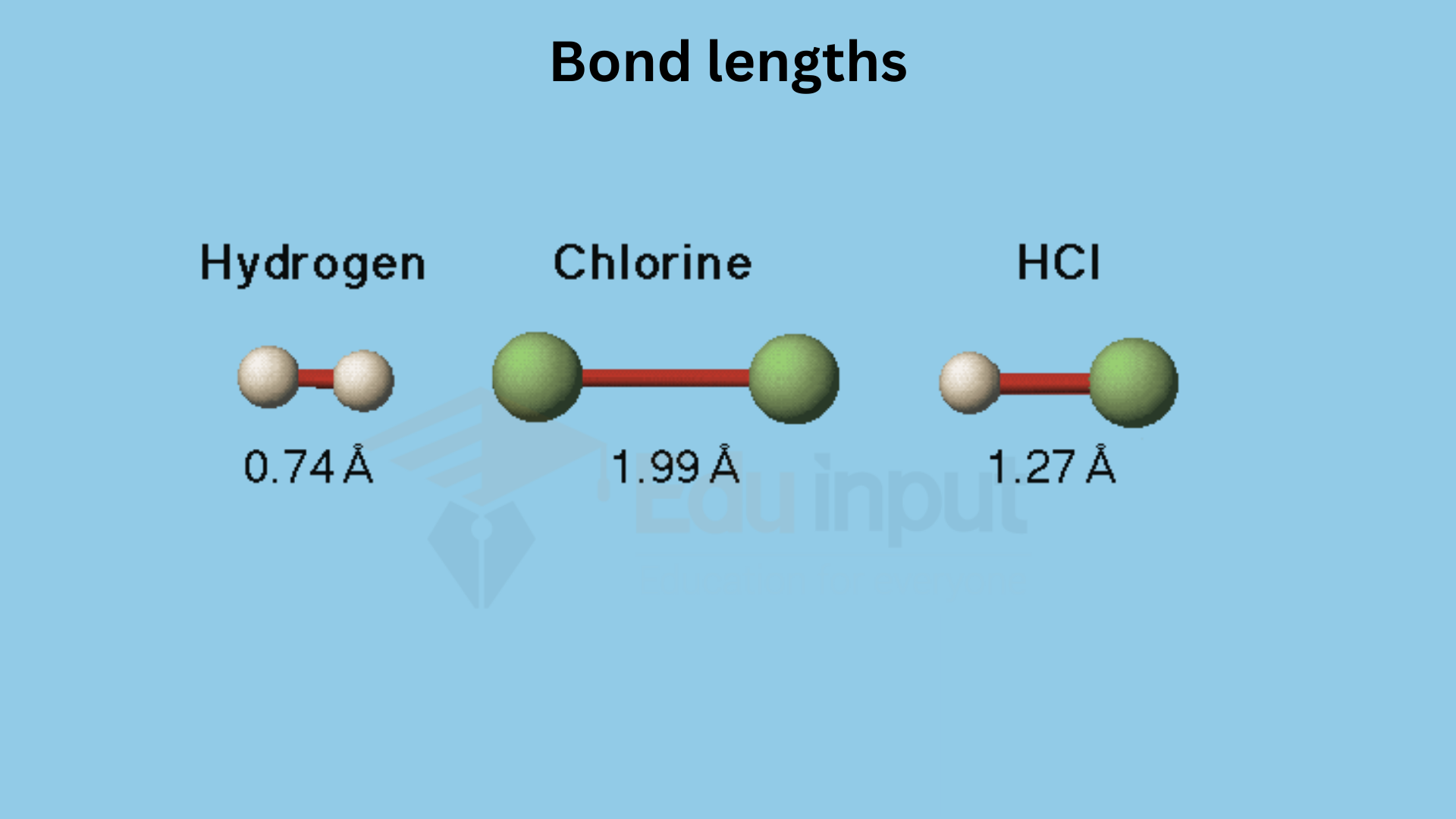
Bond length, definition, trend, factors and applications

Patai S., Rappoport Z. (Eds.) - The Chemistry of Organic Selenium and Tellurium Compounds. v.1, PDF, Functional Group
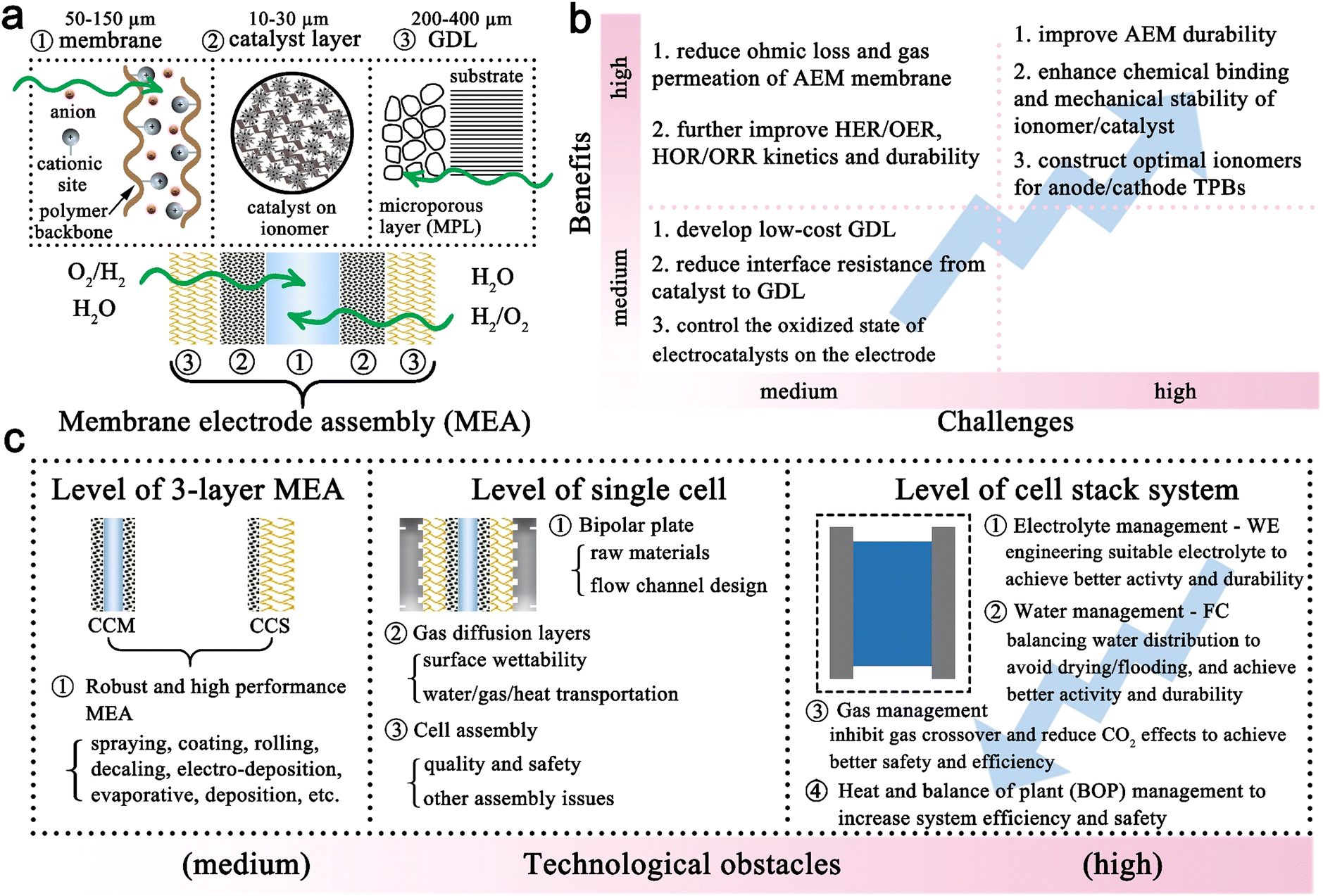
Anion-exchange membrane water electrolyzers and fuel cells - Chemical Society Reviews (RSC Publishing) DOI:10.1039/D2CS00038E
Malayalam] The electronegativities of H and F are 2.1 and 4.0 respect

Chapter 8 – Covalent Bonding - ppt download
Which oxygen-oxygen bond length is greater: H2O2 or F2O2? - Quora

Chapter 1-5 PDF, PDF, Neutron

6 S.P. - C-4.6 Calculate the theoretical value of bond length in 1. & and are 0.37A and 0.728 respectively. Flectronegativities of P & Hare 20 and 2.1 respectively.

The X-X bond length is 1.5 Å and Y-Y bond length is 1.48 Å.If electronegativity values of X and

Bansal classes chemistry study material for iit jee by S.Dharmaraj - Issuu

Atkins Physical Chemistry 10th Solutions, PDF, Gases

Inorganic Chemistry For The JEE Mains and Advanced by K Rama Rao, PDF, Atoms

Phosphorescent organic light-emitting devices: Iridium based emitter materials – An overview - ScienceDirect