
Solved P2.26 A vessel containing 2.00 mol of an ideal gas
4.6
(98)
Write Review
More
$ 31.99
In stock
Description

Answered: What is the volume of 2.00 moles of…
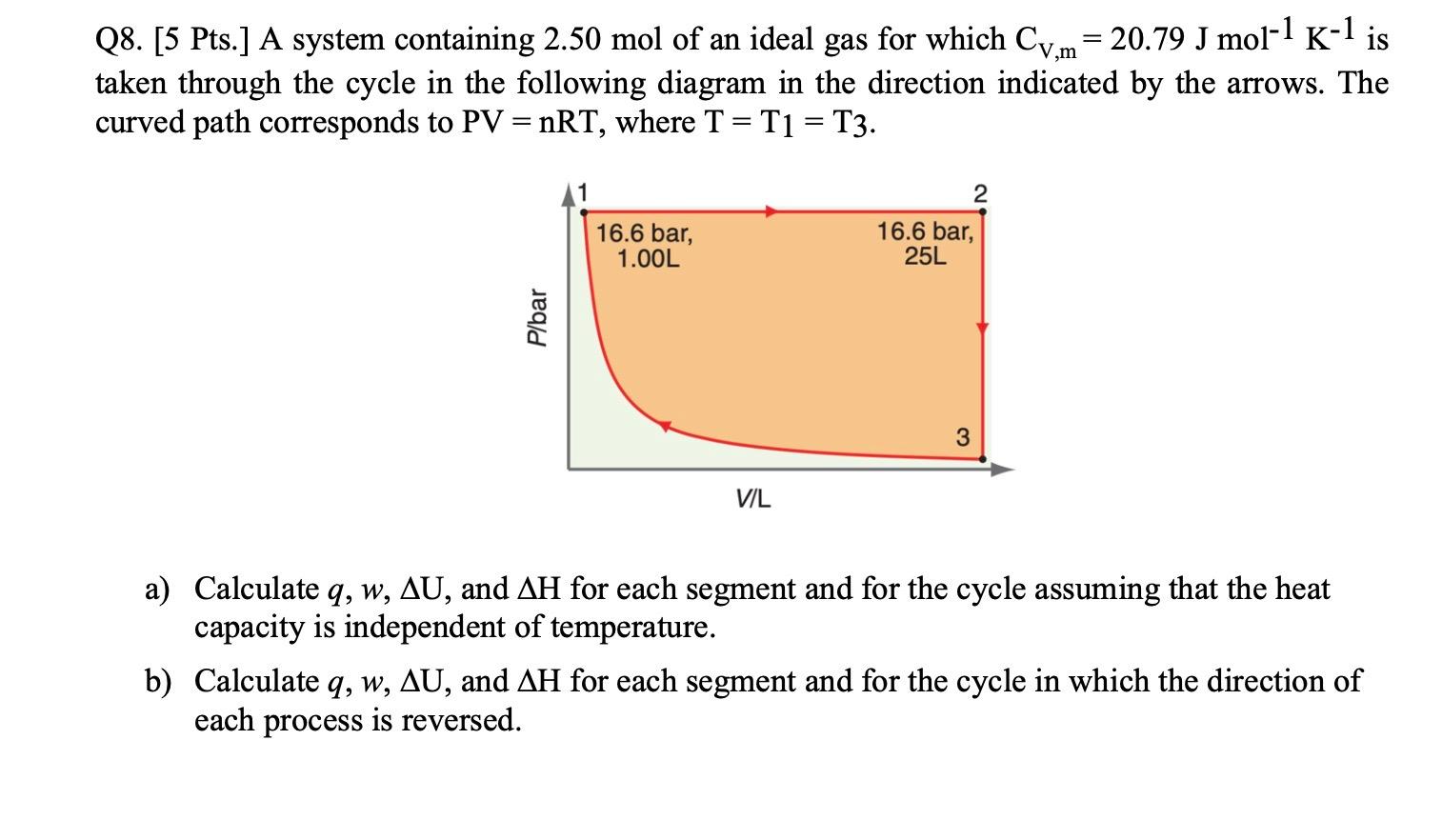
Solved Q8. [5 Pts.] A system containing 2.50 mol of an ideal

Introduction to Chemical Engineering Thermodynamics Solution Manual - Flipbook by Oya FX Trading & Investments

Answered: At 45.0 °C, a 14.00 L vessel is filled…

Lab for January 13: the ideal-gas law

To an evacuated vessel with movable piston under external pressure of 1 atm 0.1

Thermodynamics: An Engineering Approach - 5th Edition - Part I by

An ideal gas expands according to the laq PV^(3)/(2) = constant. Then

At 27^@ C, a closed vessel contains equal weights of helium (mol. wt.

Answered: A 2.00 L flask is filled with propane…
Related products
You may also like









