
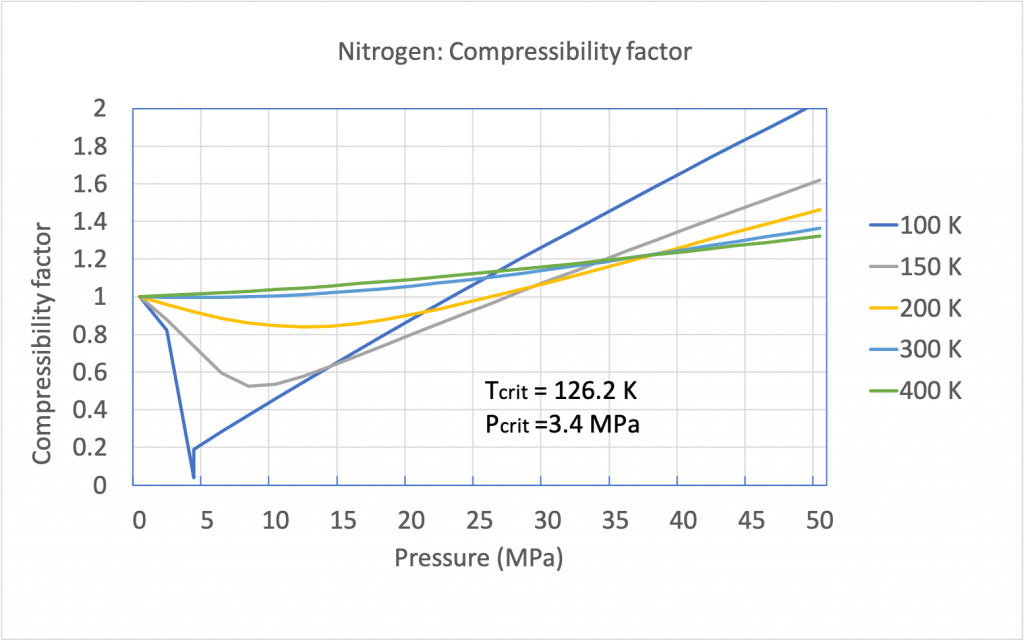
2) 1:12:15 (3) 12:15: Jals (4) 2 5 The compressibility factor nitrogen 330 K and 800 atm is 1.90 and 200 atm is 1.10.A certain mass of Noccupies a volume of 1
Click here:point_up_2:to get an answer to your question :writing_hand:2 112153 1215 jals 42 5the compressibility factor for nitrogen at 330 k and 800
Click here👆to get an answer to your question ✍️ -2- 1-12-15 -3- 12-15- Jals -4- 2 5 The compressibility factor nitrogen 330 K and 800 atm is 1-90 and 200 atm is 1-10-A certain mass of Noccupies a volume of 1 dmat 330 Kand eoo atm calculate volume occupied by same cuany of gas 750 K and 200 atm- -1- 1 L -2- 2L -3- 3L
3.3: Real gas and compressibility factor - Engineering LibreTexts

At 21.5^@C and a total pressure of 0.0787 atm, N_2O_4is 48.3

The compressibility factor for nitrogen at 330K and 800 atm is 1.90 an

Solved You are given the following data for the

SOLUTION: Fluid mechanics problem exercises chapter 1 - Studypool

Telugu] {:(Column-I ,Column-II ),(A) Hydrogen gas (P = 200 atm

Advanced Thermodynamics Note 1 The 1st law and other basic

Compression Factor Exam Problem using Molar Volumes - Fully
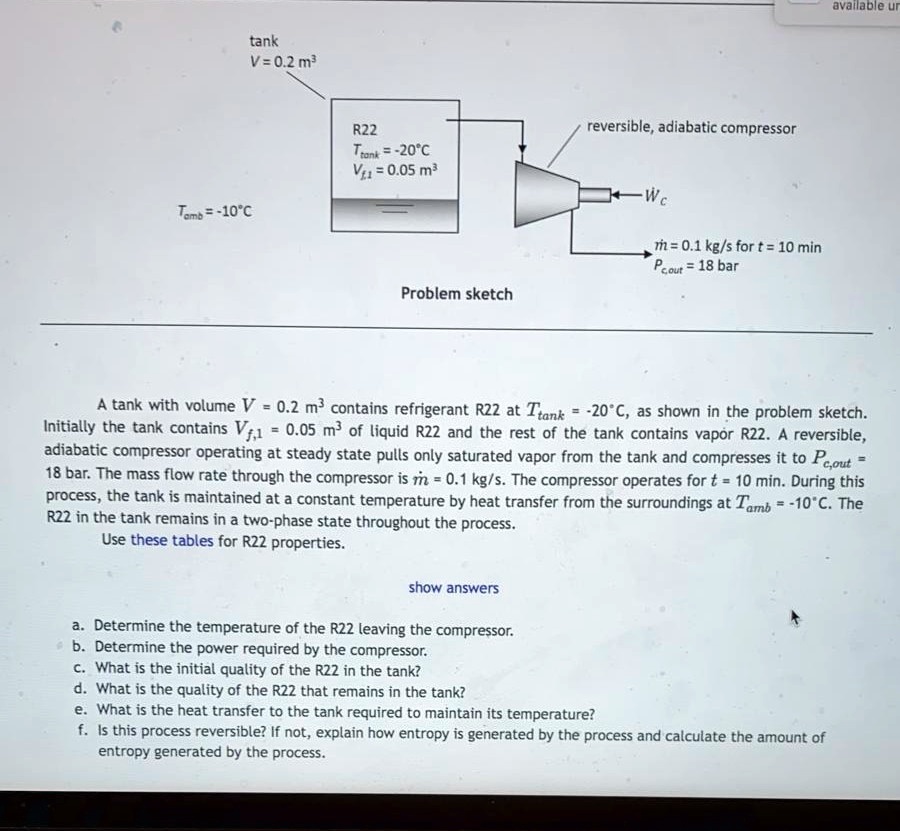
SOLVED: Text: available u tank V=0.2 m^3 R22 Tan=-20°C V=0.05 m^3

The compressibility factor of N_2 330 K and 800 atm is 1.90 and

Solved 2.86 Find the compressibility factor for nitrogen at

PDF) FluidMechWhite5eCh09.pdf Luciano Teixeira de Morais









