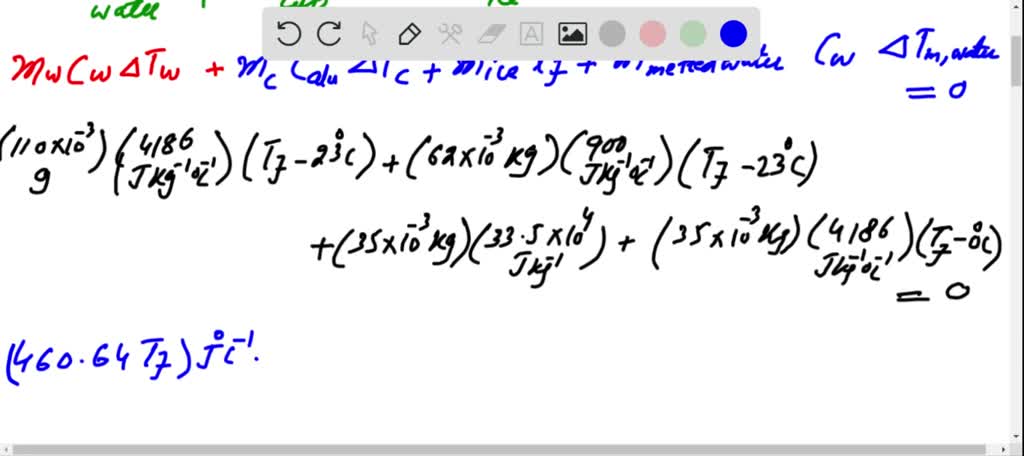
Solved Problem 3 (Phase changes) A 35 g ice cube at 0°C is
4.7
(645)
Write Review
More
$ 14.00
In stock
Description
Answer to Solved Problem 3 (Phase changes) A 35 g ice cube at 0°C is

14.24 A 0.0500-kg ice cube at −30.0ºC is placed in 0.400 kg of 35.0ºC water in a very
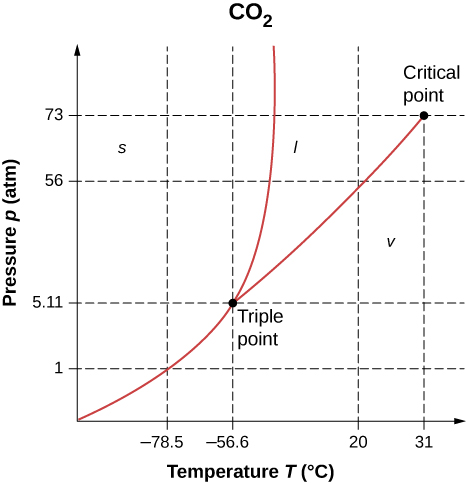
1.5 Phase Changes – University Physics Volume 2

Solved A 53.3-g ice cube is initially at 0.0°C. (a) Find the
:max_bytes(150000):strip_icc()/IceToSteam-58d96a7c3df78c516242a8cc.jpg)
Calculate Energy Required to Turn Ice Into Steam

Two 50 gm ice cubes are dropped into 250 gm of water into a glass. If the water was initially a temperature of 25^{0}C and the temperature of ice - 15^{0}C. Find
How much energy is required to change a 35-g ice cube from ice at -25 degrees C to steam at 115 degrees C? - Quora
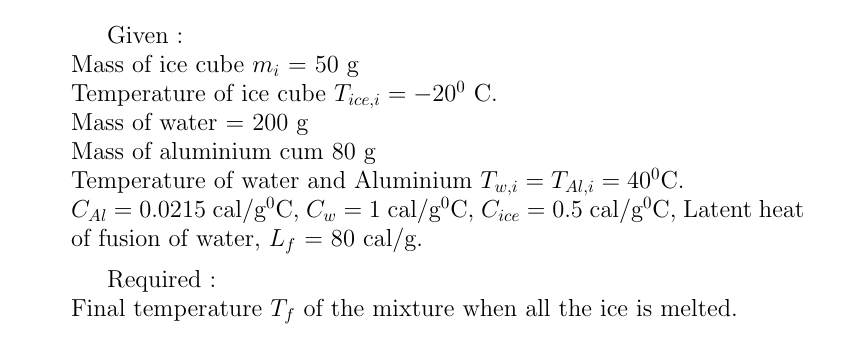
Answered: A 50 g ice cube, initially at -20degree…

3.4 Solving Energy Problems Involving Phase Changes and Temperature Changes

Related products
You may also like