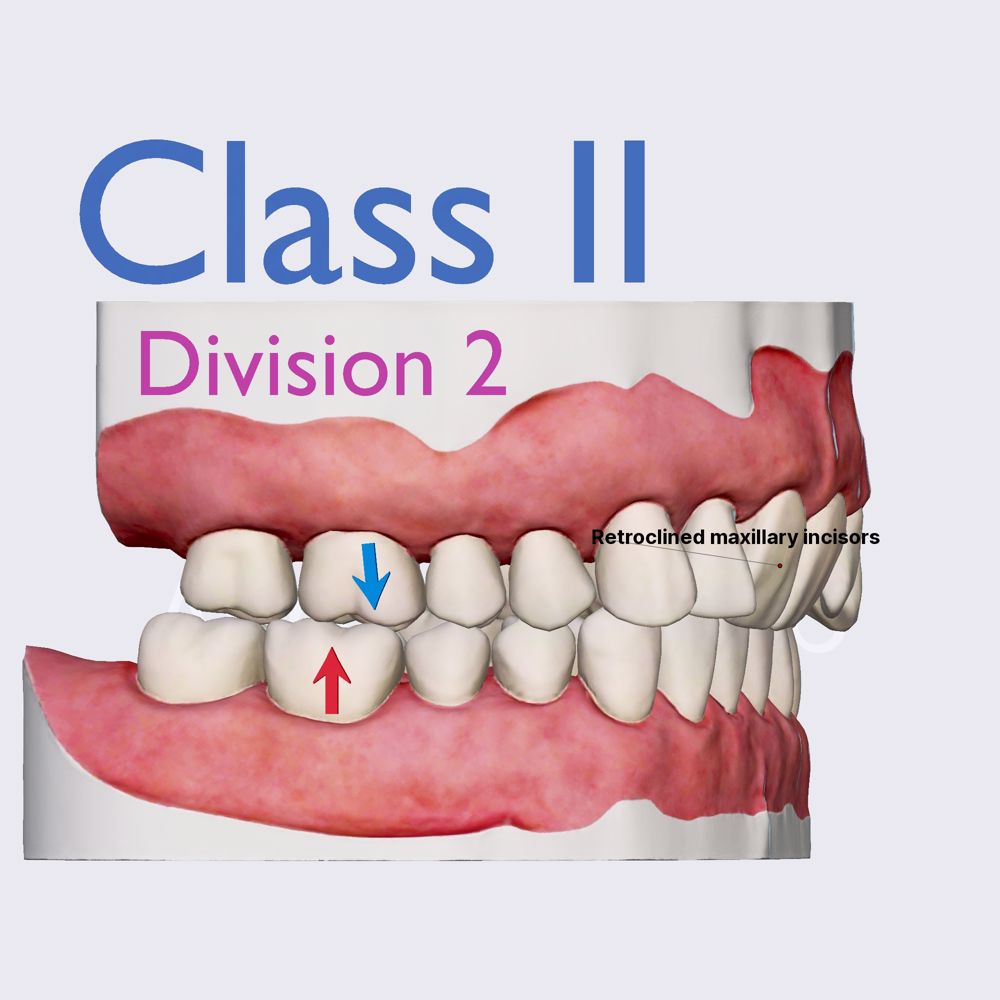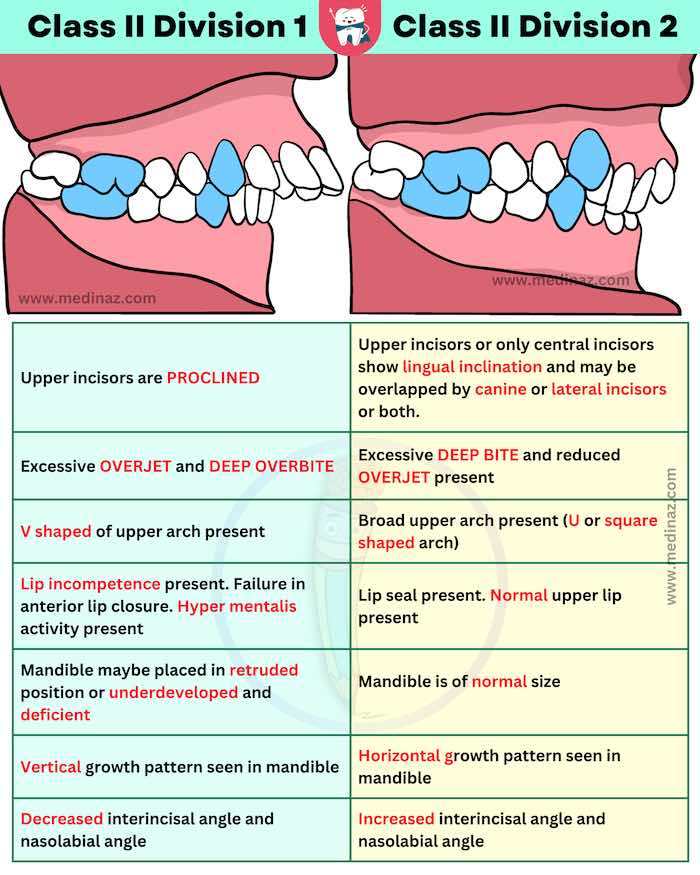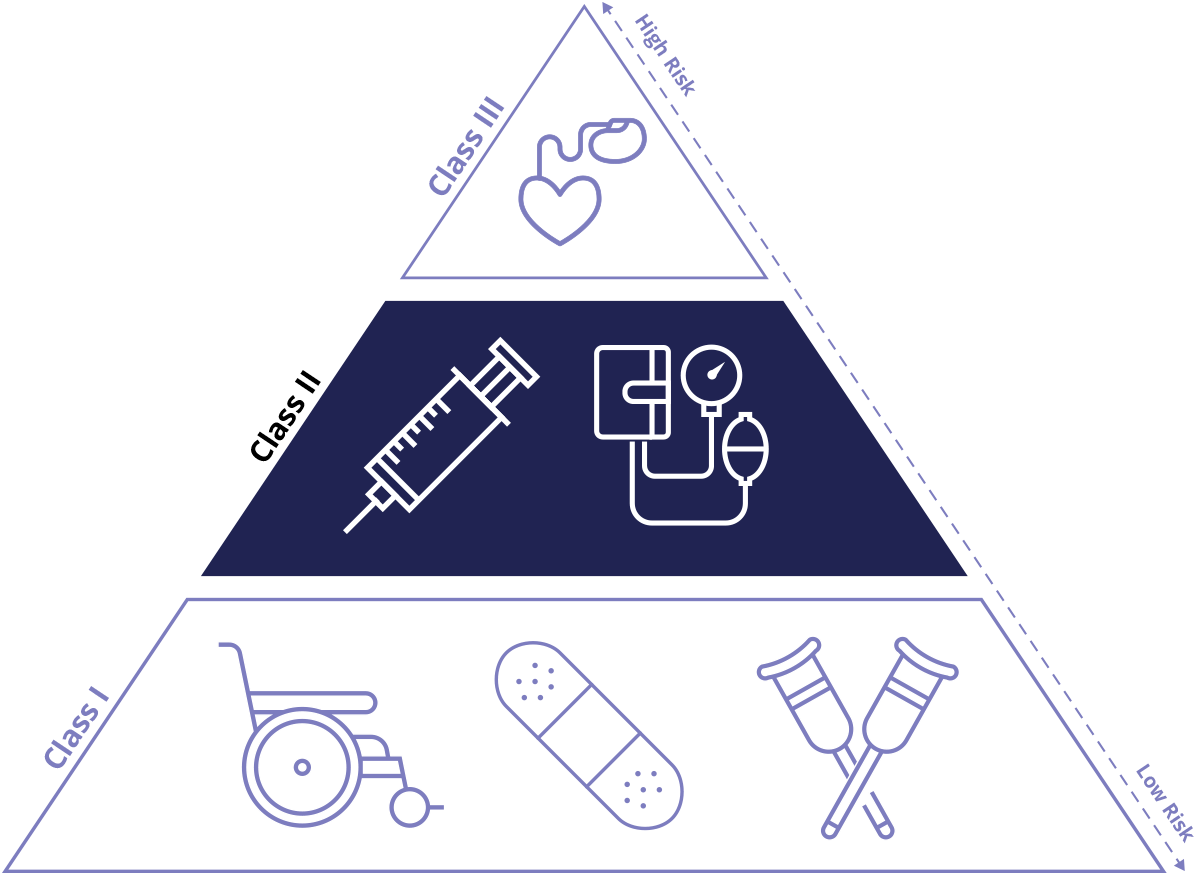
Class II Device Definition
Class II medical devices have moderate to higher risks to patients or users. Over 40% of medical devices fall into this device category. The majority of medical devices are considered to be Class II devices. Some examples of Class II devices include catheters, syringes, contact lens, and pregnancy test kits.
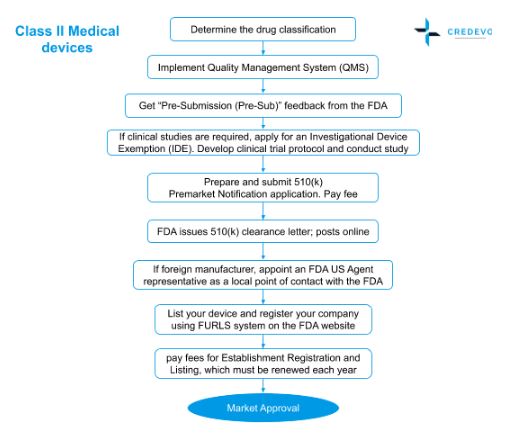
Medical Device Market Approval Process in the United States

De Novo classification process: a beginner's guide

The 3 FDA medical device classes: differences and examples explained

What's the Difference between a Class I Medical Device and a Class II?

What's the Difference between a Class I Medical Device and a Class II?

What's the Difference between a Class I Medical Device and a Class II?

How to Determine Your Medical Device's FDA Classification

Medical device regulations, classification & submissions

Understanding FDA Device Classes Infographic

What's the Difference between a Class I Medical Device and a Class II?
Medical Device Classification Guide - How To Determine Your Device Class
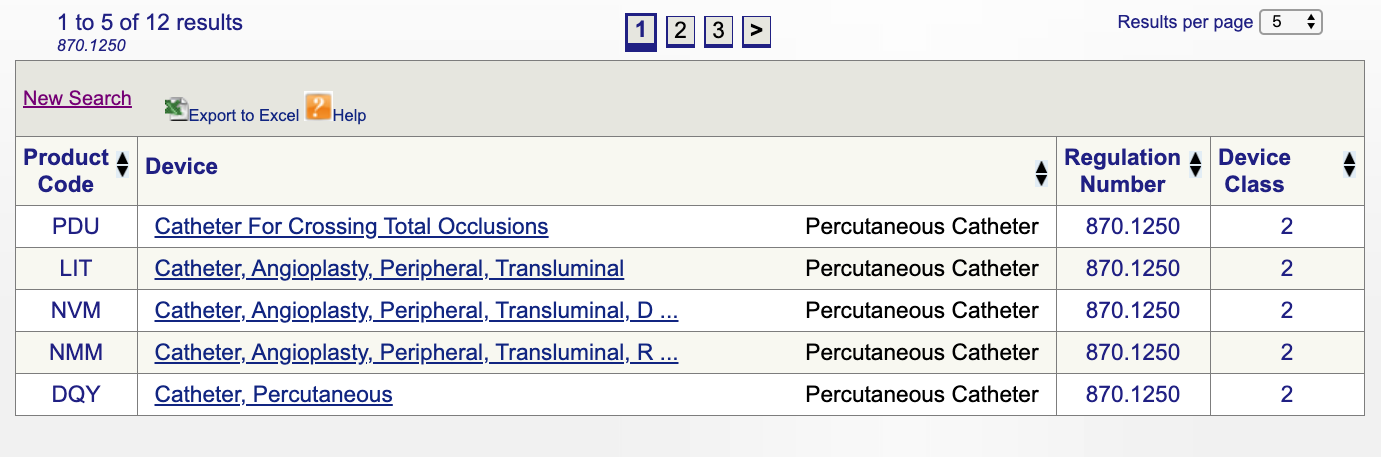
Medical Device Classification Guide - How To Determine Your Device Class
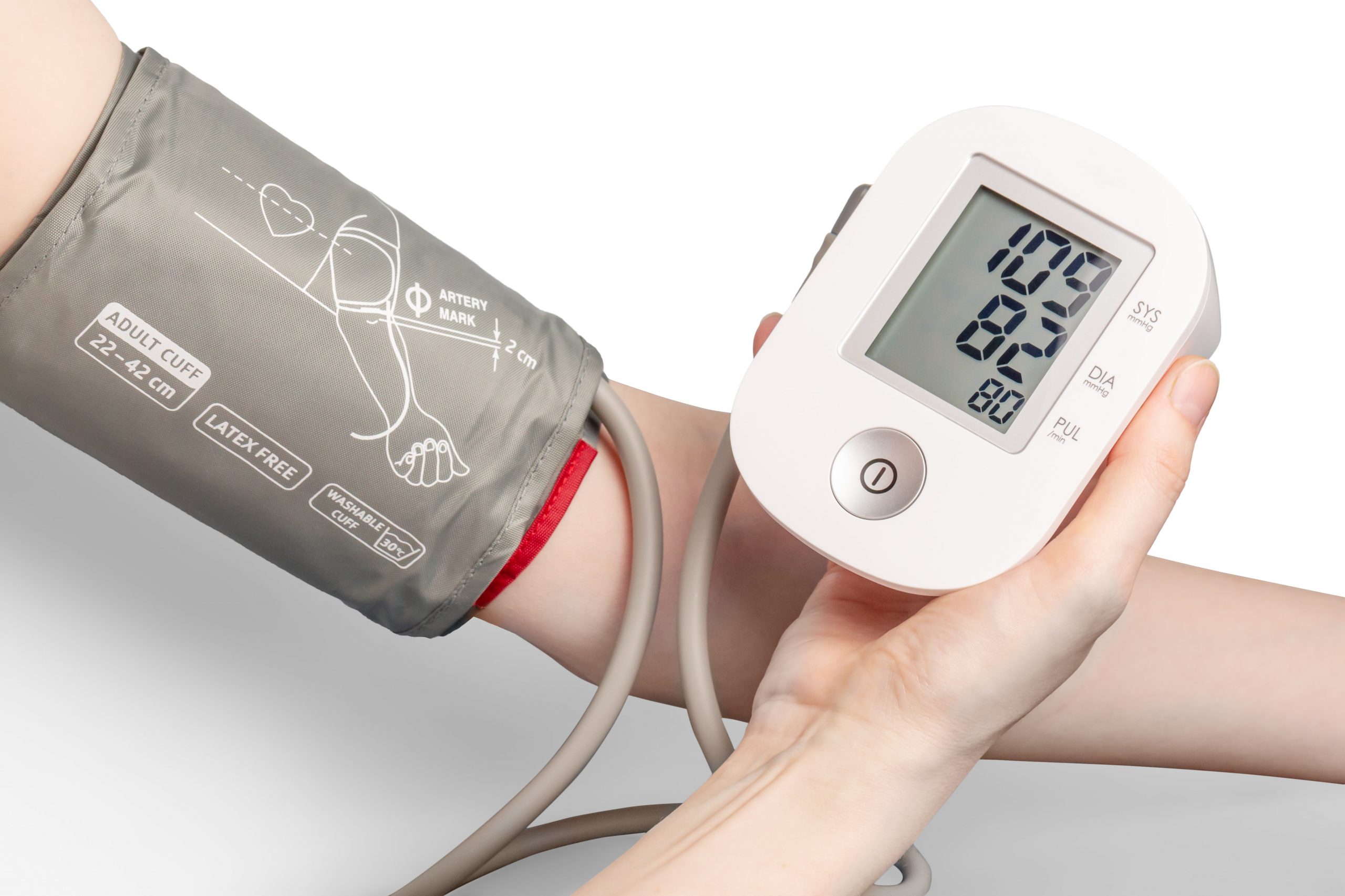
What is a class II medical device?
Implantable medical devices