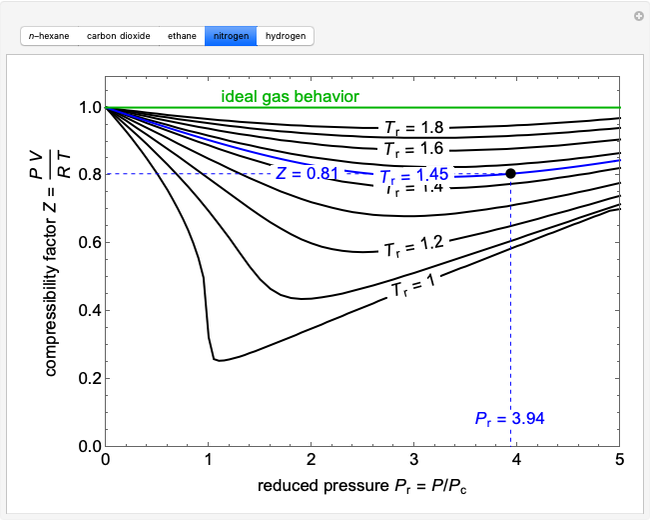
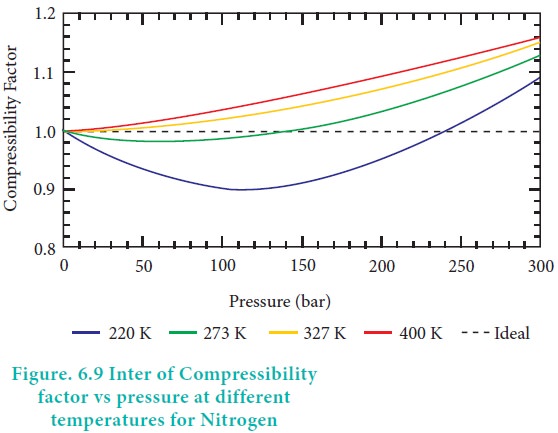
Compressibility factor Z = PV / nRT is plotted against pressure as
Compressibility factor Z = PV / nRT is plotted against pressure as shown below:What is the correct order for the liquefiability of the gases shown in the above graph? A. CO 2
Compressibility factor Z - PV - nRT is plotted against pressure as shown below-What is the correct order for the liquefiability of the gases shown in the above graph- A- CO 2- CH 4- N 2- H 2B- H 2- CH 4- N 2- CO 2C- CH 4- H 2- N 2- CO 2D- H 2- N 2- CH 4- CO 2
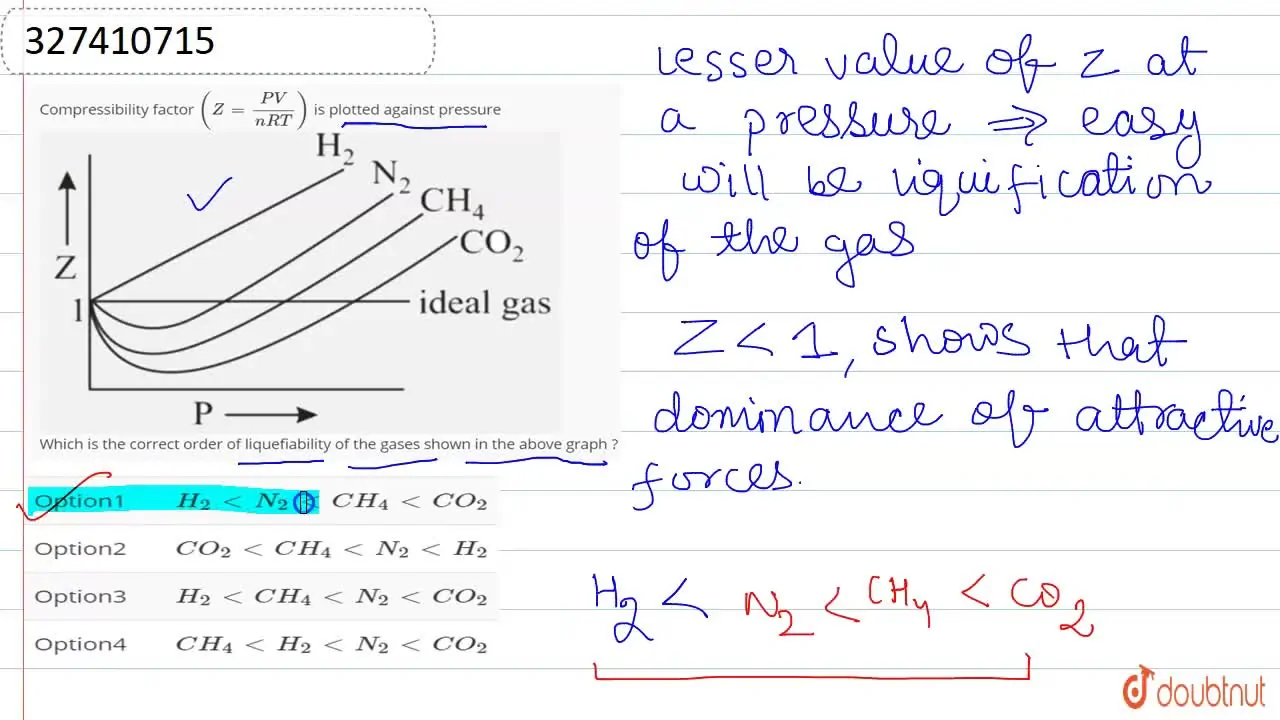
Compressibility factor (Z=(PV)/(nRT)) is plotted against pressure

Compressibility factor (Z=(PV)/(nRT)) is plotted against pressure

Van der Waals equation - Wikipedia

Deviations from ideal gas behaviour, intermolecular forces, Van der Waals equation of state, compressibility factors and the critical pressure and critical temperature of a gas revision notes doc brown's chemistry UK advanced

Gas Compressibility - an overview

A real gas M behaves almost like an ideal gas. Graph 1 is obtained by plotting volume, V against temperature, T for x mol of gas M at pressure, P_1. a. Suggest

gas laws - Does the amount of a gas increase with pressure? - Chemistry Stack Exchange

shows plots of the compressibility factor, Z = P V /RT , of methanol at

Compressibility factor (Z) is plotted against pressure at different temperature for same gasWhic
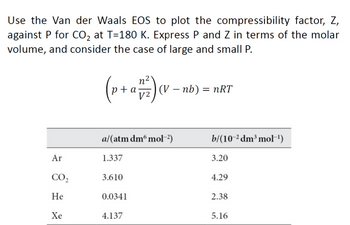
Answered: Use the Van der Waals EOS to plot the…
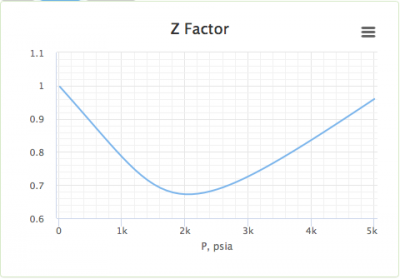
Finding the compressibility factor (Z)

Non-Ideal Gas Behavior