Energies, Free Full-Text
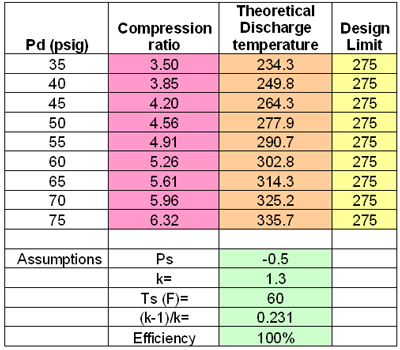
Understanding how the pressure level affects the efficiency of liquid piston gas compression is essential for a greater applicability of the technology in compressed air energy storage. To explore the impacts, compression starting at three different initial pressure levels (1, 2, 3 bar) with a pressure ratio of 2 is performed, and how isothermal compression efficiencies are affected depending on the initial pressures is analyzed. Under the experimental conditions, higher initial pressure leads to lower isothermal efficiency. Air dissolution during the compression is also investigated because the chamber is a pressure-varying and a liquid-containing environment, where the gas solubility changes during the process. Evaluating the dissolution is critical as it affects the energy output when the compressed air is expanded to regenerate the energy. The changes in the air mass and the retrievable volume of the air after expansion are quantified based on Henry’s law. For a compression at higher pressure, because the air solubility is proportional to pressure, a greater reduction in the air mass and volume percentages is expected. This trend of the mass decreasing with the pressure level leads to less energy output than the originally intended output when the stored energy is retrieved in a discharging process.

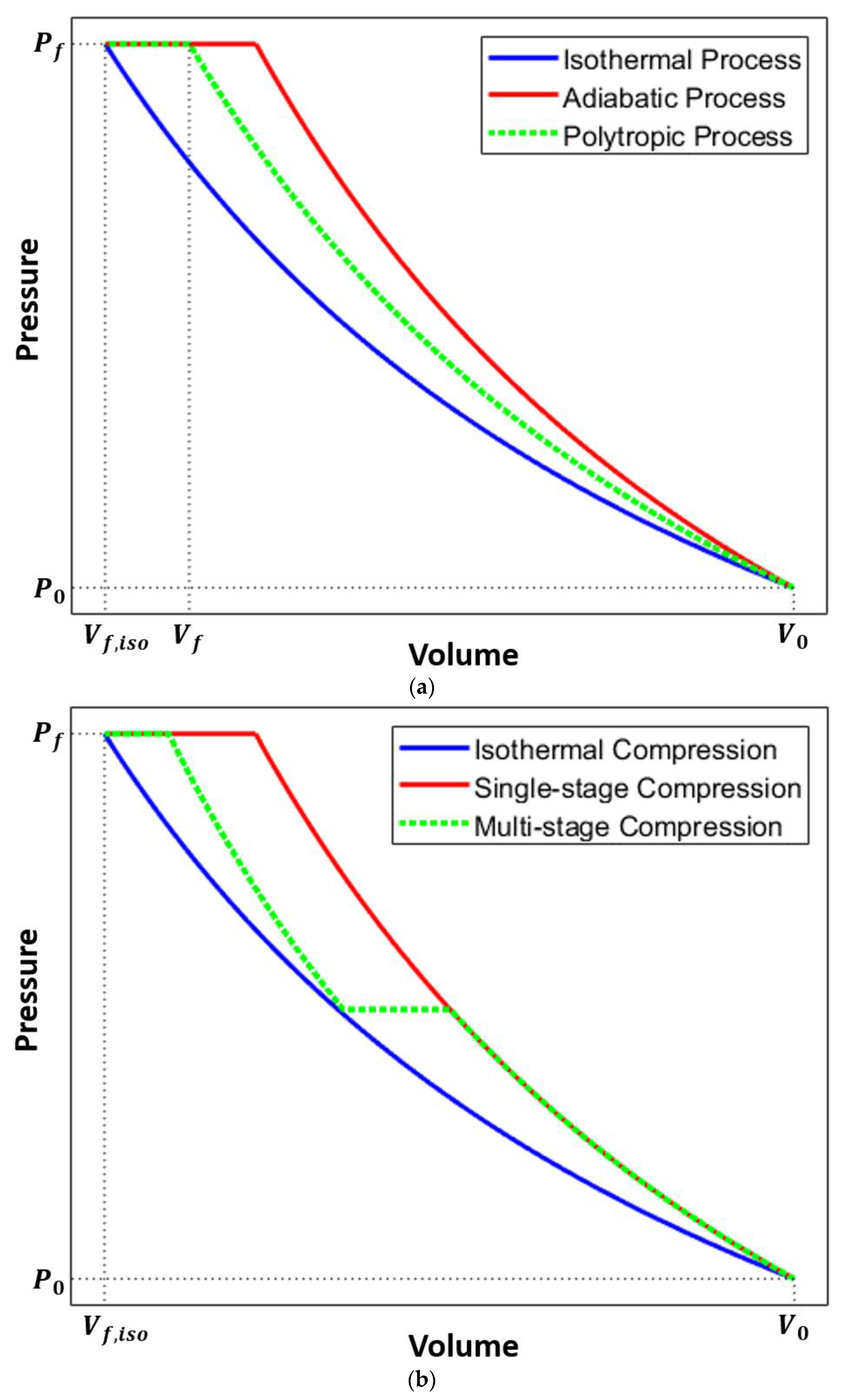
The diagram shows that the effective Gibbs free energy decreases with

Energies, Free Full-Text, alpha zero campinas

Energies, Free Full-Text, alpha zero campinas

All Energy Australia Exhibition & Conference

2 Minute Energy Clearing Ritual - The Awakened Professional
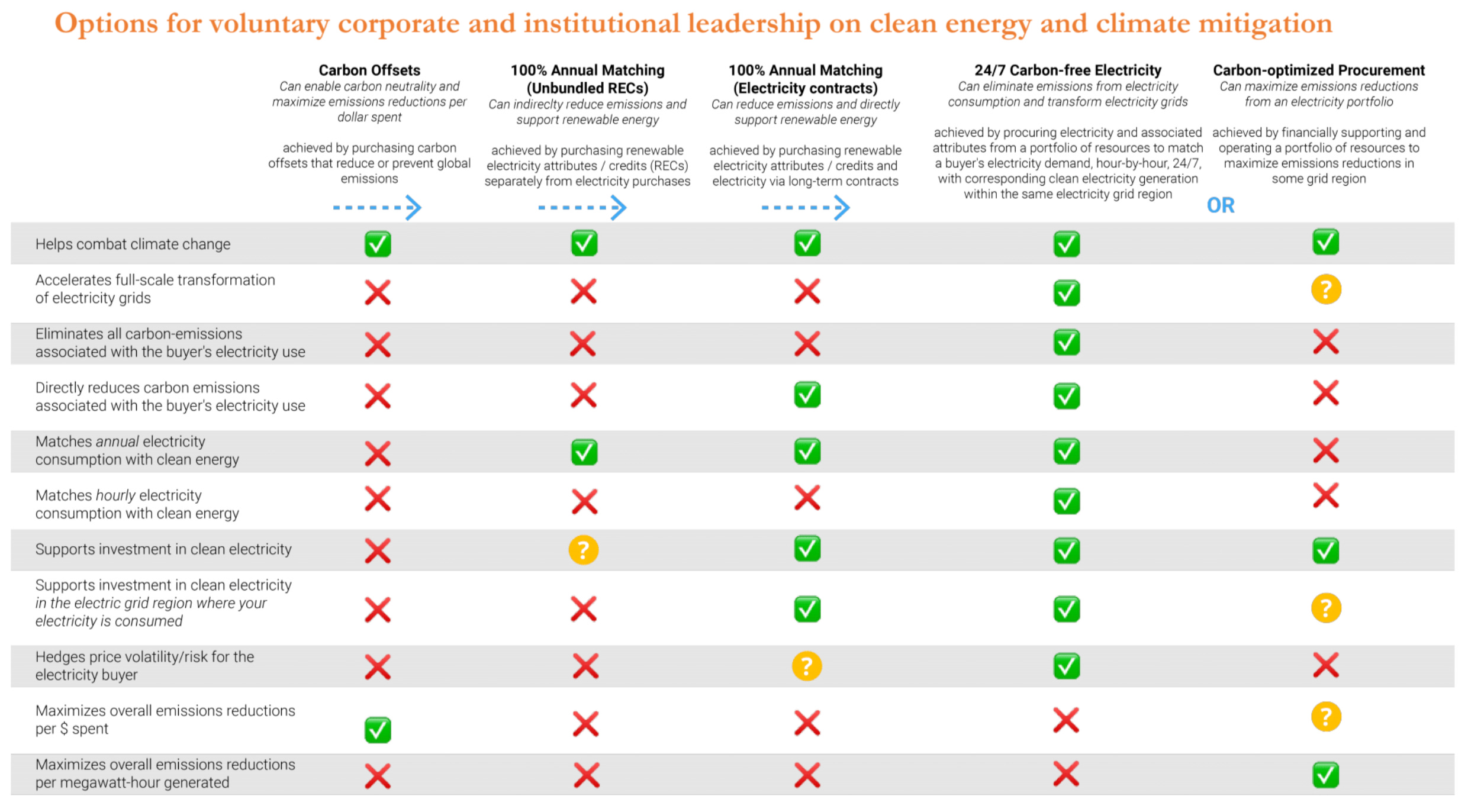
Energy systems and 24/7 carbon free energy

24/7 Carbon-Free Energy: Methods, Impact & Benefits

Energies, Free Full-Text, alpha zero campinas
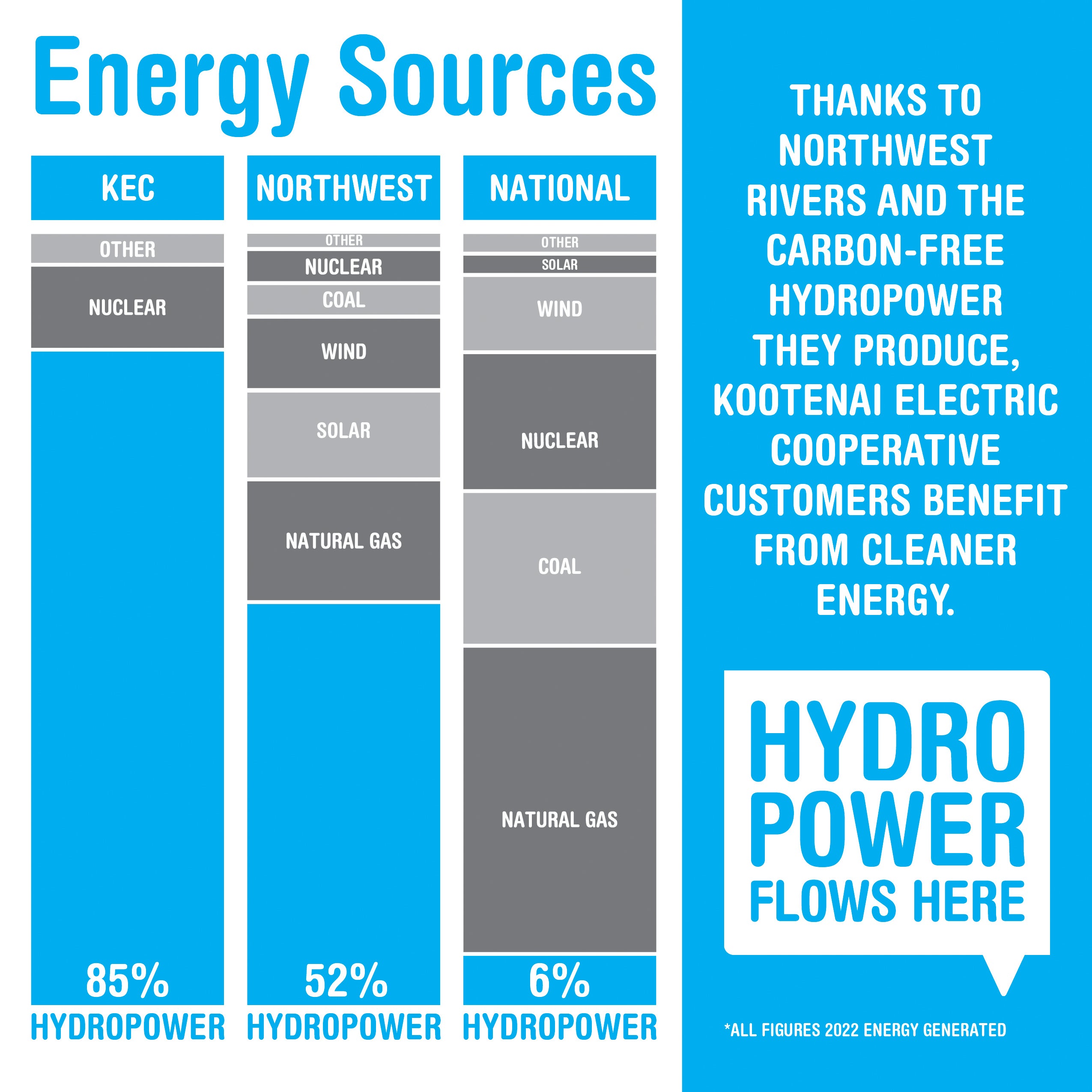
Carbon Free Energy Options Kootenai Electric Cooperative

Inner pore hydration free energy controls the activation of big potassium channels - ScienceDirect

Top panel-convergence of the total free energy E in eV for the RMM-DIIS

Energy Infobooks – The NEED Project

Living Systems Requirement for Free Energy and Matter - Lesson