Solved Show that the compressibility factor of van der Waals
Answer to Solved Show that the compressibility factor of van der Waals

Van der Waals equation - Wikipedia

09 DEFINITION Behaviour of gases by van der Waals equation (P+*}(0-b) = RT may be written as (P+*}() =RT of PV + 9 =RT of PV=RT - For large V (at very
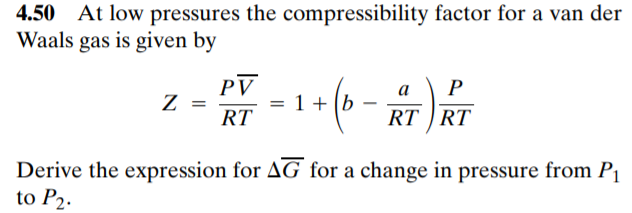
Solved 4.50 At low pressures the compressibility factor for
Van der waals equation: Derivation, Explanation

Solved 3) The compressibility factor for a real fluid with
If Z is a compressibility factor, van der Waals' equation at low pressure can be written as - Sarthaks eConnect

What is the major factor that accounts for most of the difference in these two values of pressure (ideal gas law vs. van der Waals equation)?

Non-Ideal Gas Behavior Chemistry: Atoms First

Is there a set of conditions at which the compression factor

Behaviour of Real Gases, PDF, Gases