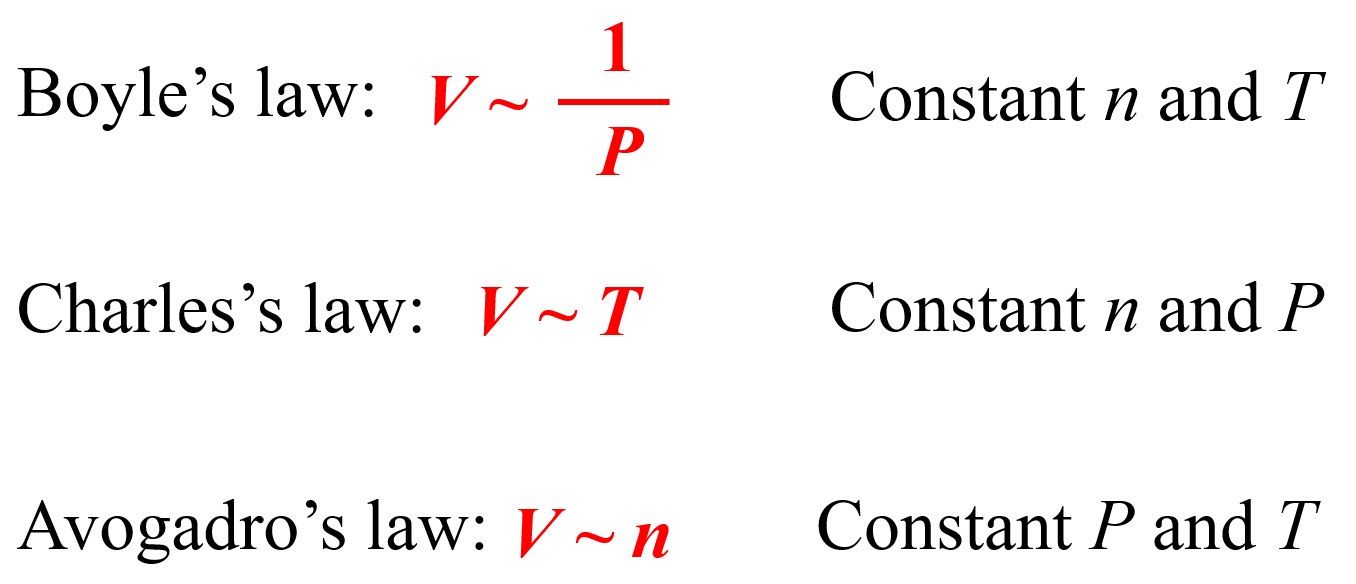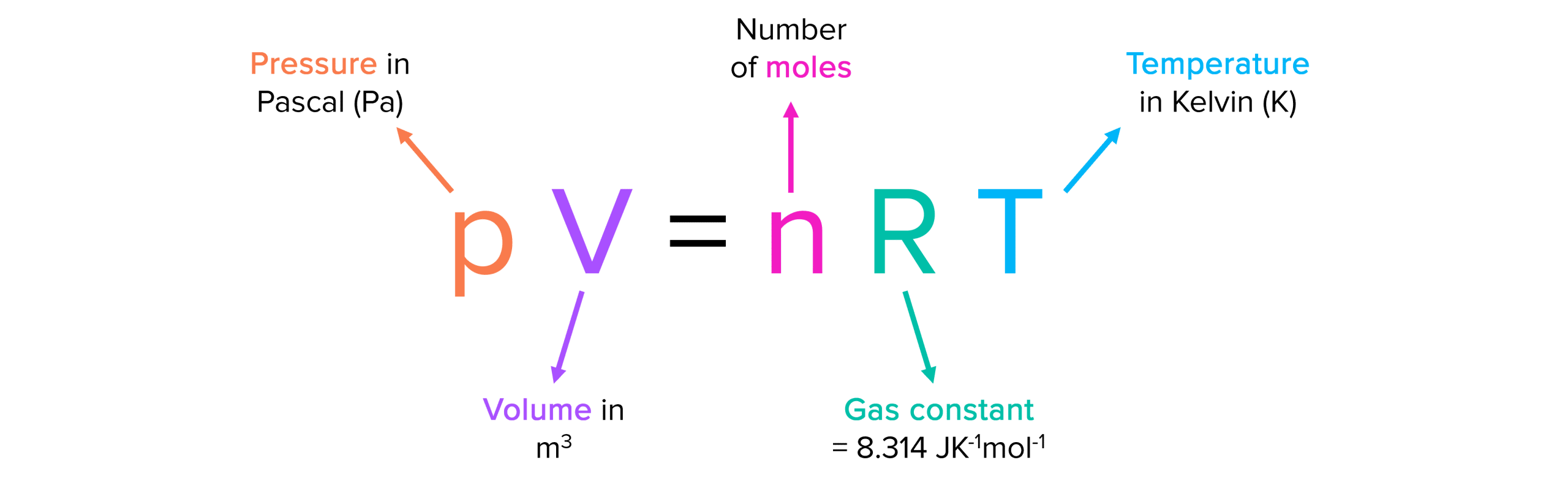kinetic theory - Why doesn't Helium behave as an ideal gas? - Physics Stack Exchange
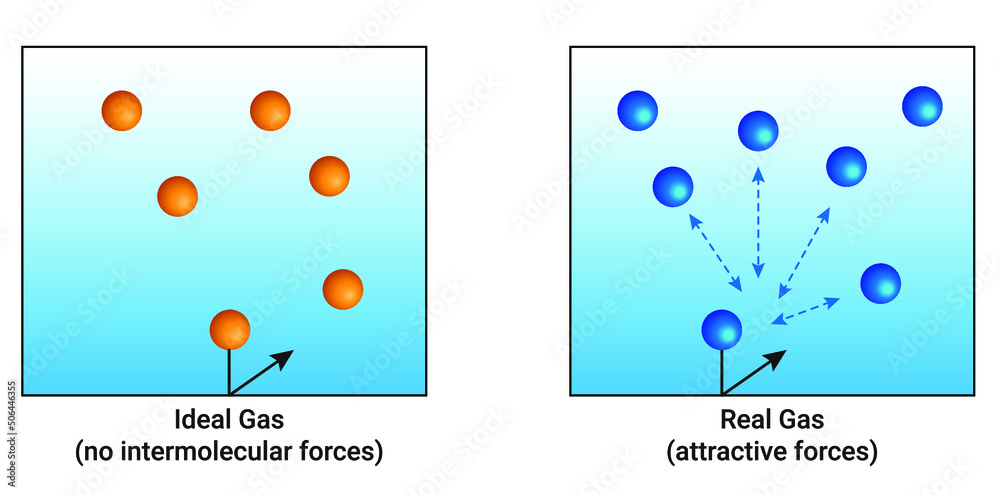
I am a bit confused (might be due to some conceptual misunderstanding) as to why doesn't Helium behave as an ideal gas (it shows a deviation from the $pV$ vs $p$ graph)? (Given the fact that it is
Is helium the noble gas that acts most like an ideal gas? - Quora

Lecture 18 Chapter 18 Kinetic Theory of Gases - ppt download
What is an intuitive explanation for the fact that the Boltzmann distribution of energies in a gas is independent of mass while the distribution of velocities is not? - Quora
Why can't mass be converted into energy? - Quora
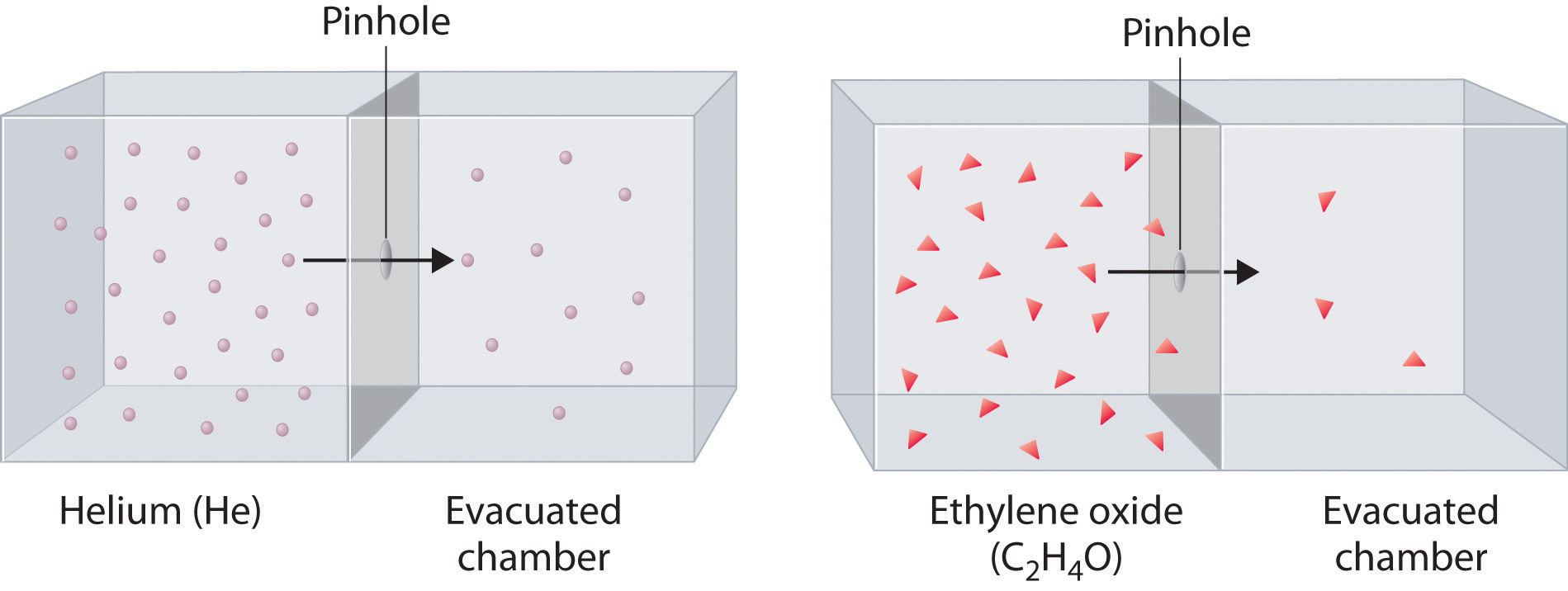
The Kinetic Molecular Theory of Gases

What Is the Universe Made of? (Matter, Energy, and Interactions)

download - Enea
What are alpha particles? Do they have a charge or mass? If so, how can they pass through matter without any harm to their structure (like beta and gamma rays)? - Quora
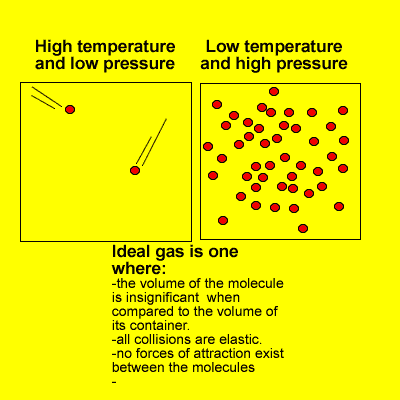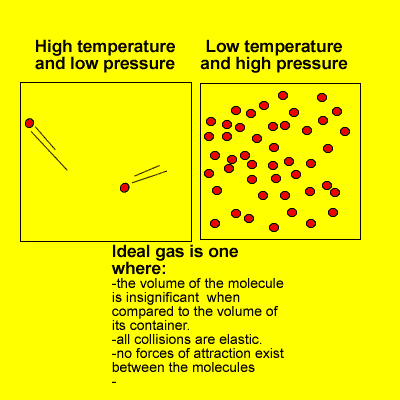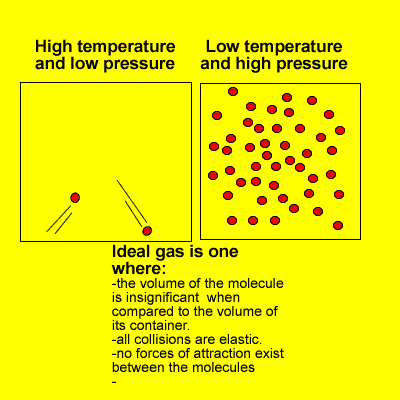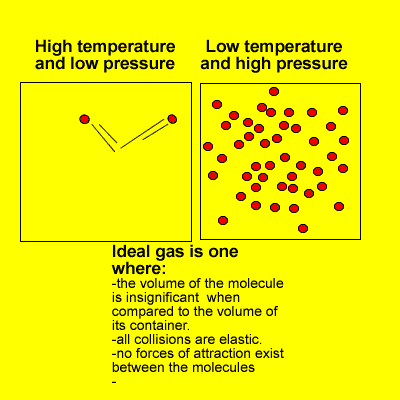
Chemistry - Ideal gas-kinetic theory of gases
Why can helium not be transformed into solid by cooling? - Quora
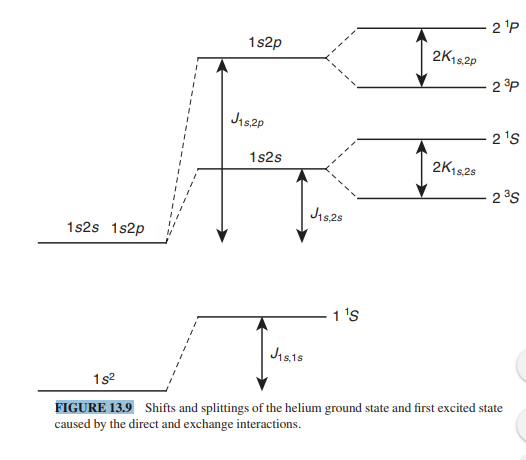
Get Answer) - Find the first-order perturbed energies of the helium excited
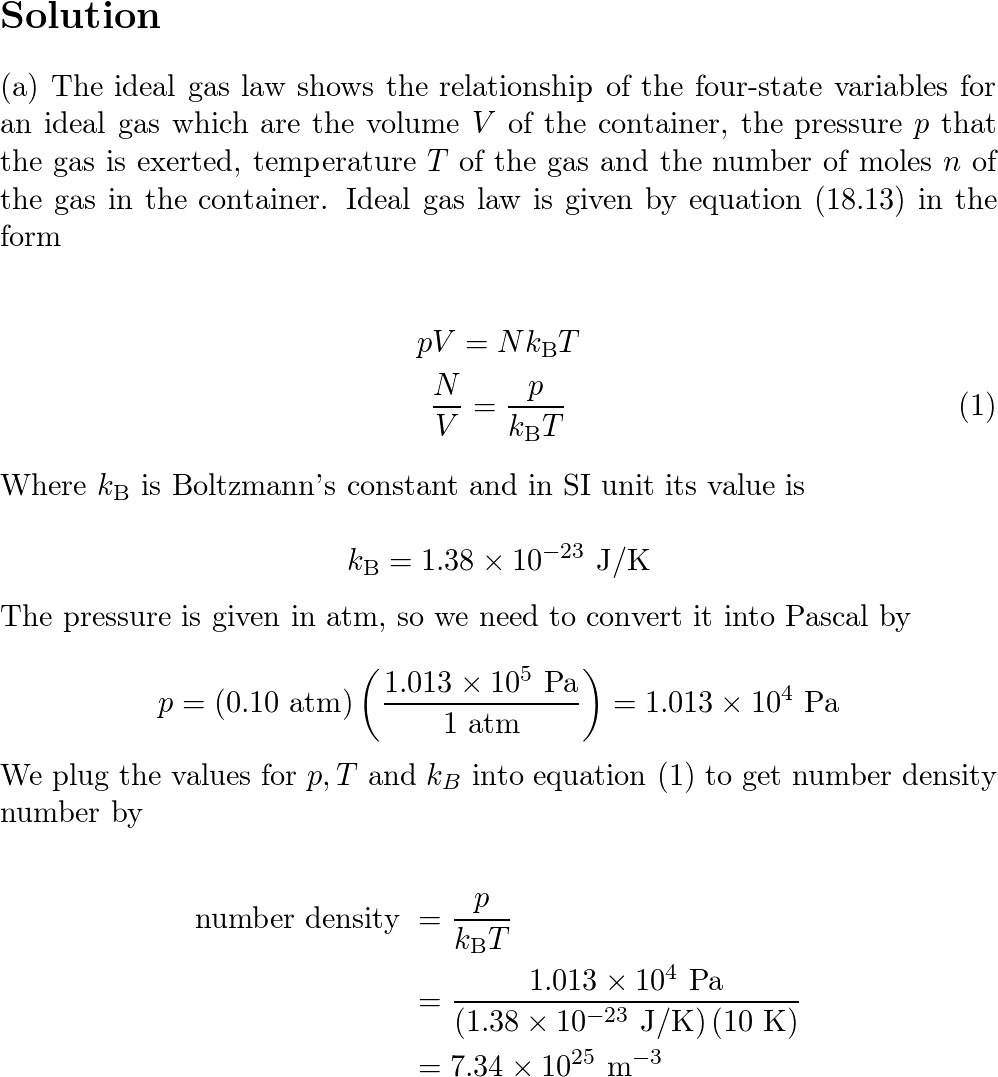
During a physics experiment, helium gas is cooled to a tempe

kinetic theory - Why doesn't Helium behave as an ideal gas? - Physics Stack Exchange