
If Z is a compressibility factor, van der Waals equation at low pressure ..
Solution For If Z is a compressibility factor, van der Waals equation at low pressure can be written as
Video solution 1: If Z is a compressibility factor, van der Waals equation at low pressure can be written as
Video solution 2: If Z is a compressibility factor, van der Waals equation at low pressure can be written as
Video solution 3: If Z is a compressibility factor, van der Waals equation at low pressure can be written as
Video solution 4: If Z is a compressibility factor, van der Waals equation at low pressure can be written as

Hey guys, What is the correct answer to this question? #study24x7
If Z is a compressibility factor, van der Waals' equation at low

SOLUTION: Dpp 7 gaseous state and chemical energetics - Studypool

If Z is a compressibility factor, van der Waals' equation at low

If Z is a compressibility factor, van der Waals equation at low

For one mole of a Van der Waals gas when b=0 and T=300 K, the PV

physical chemistry - Why do some gases have lower value of Z for a

The van der Waals equation for CH(4) at low pressure is
The compression factor (compressibility factor) for one mole of a

If Z is a compressibility factor, van der Waals equation at low
If Z is a compressibility factor, van der Waals equation at low

Compressibility factor - Wikipedia

1.7: Connecting the van der Waals and the viral equations: the
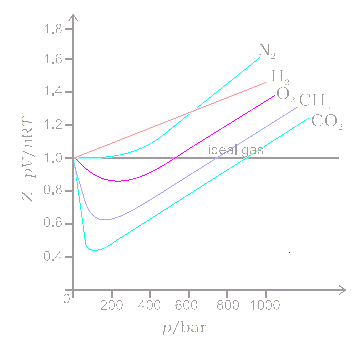
Van der waals equation: Derivation, Explanation
Van der waals equation: Derivation, Explanation

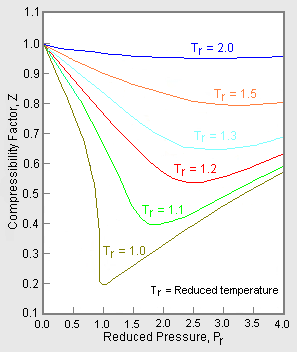





.jpg)
