Example of a primary drying design space graph showing sublimation

Feroz Jameel's research works Gilead Sciences, Foster City and

Best Practices and Guidelines (2022) for Scale-up and Technology Transfer in Freeze Drying Based on Case Studies. Part 2: Past Practices, Current Best Practices, and Recommendations

Figure A1. Product temperature for 5.4 mL and 10.6 mL fill volumes

Choked flow and importance of Mach I in freeze-drying process
Graph for primary drying design space that incorporates time as a

On the Use of Mathematical Models to Build the Design Space for the Primary Drying Phase of a Pharmaceutical Lyophilization Process - Journal of Pharmaceutical Sciences
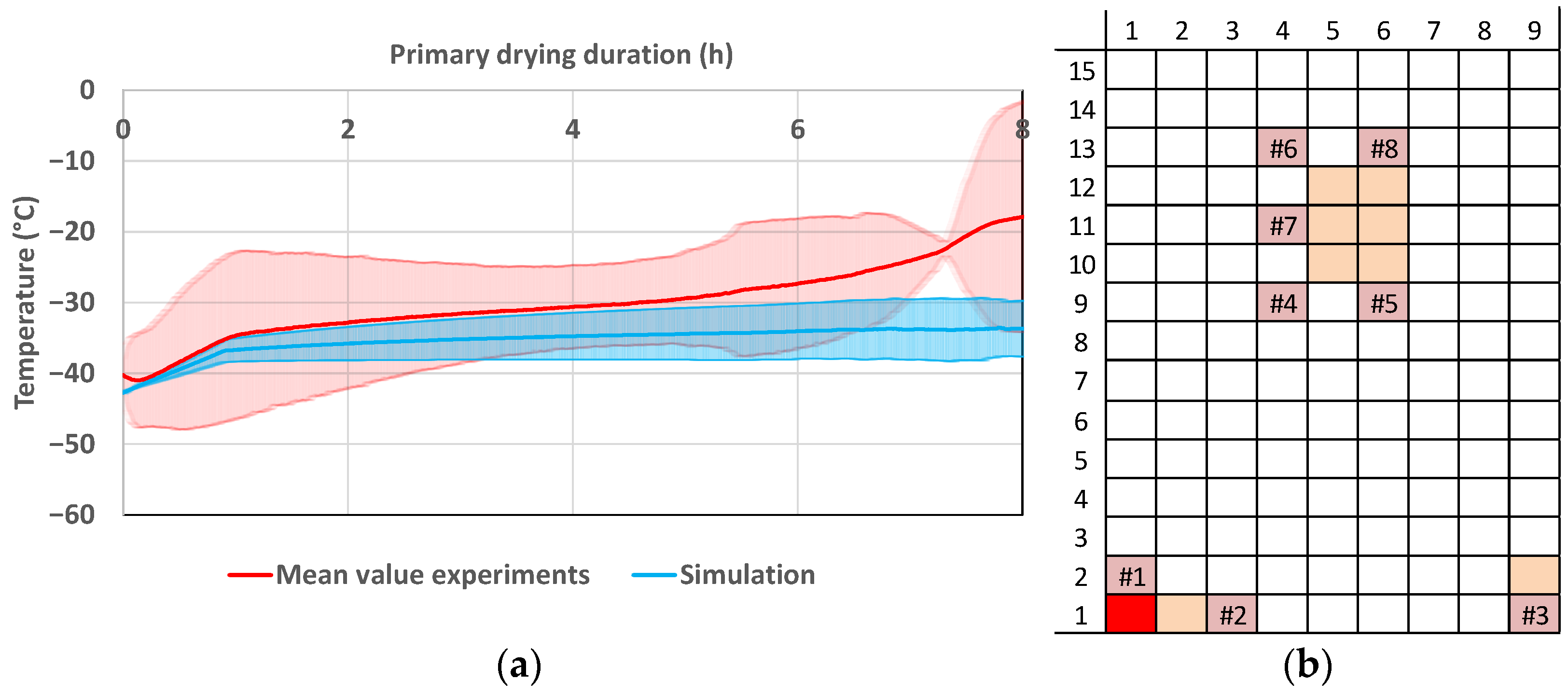
Pharmaceutics, Free Full-Text
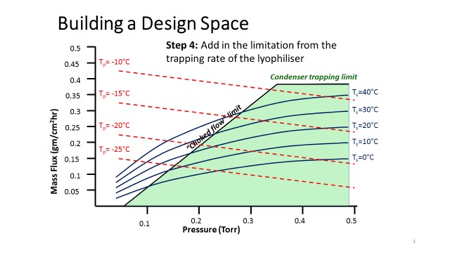
QbD in freeze drying
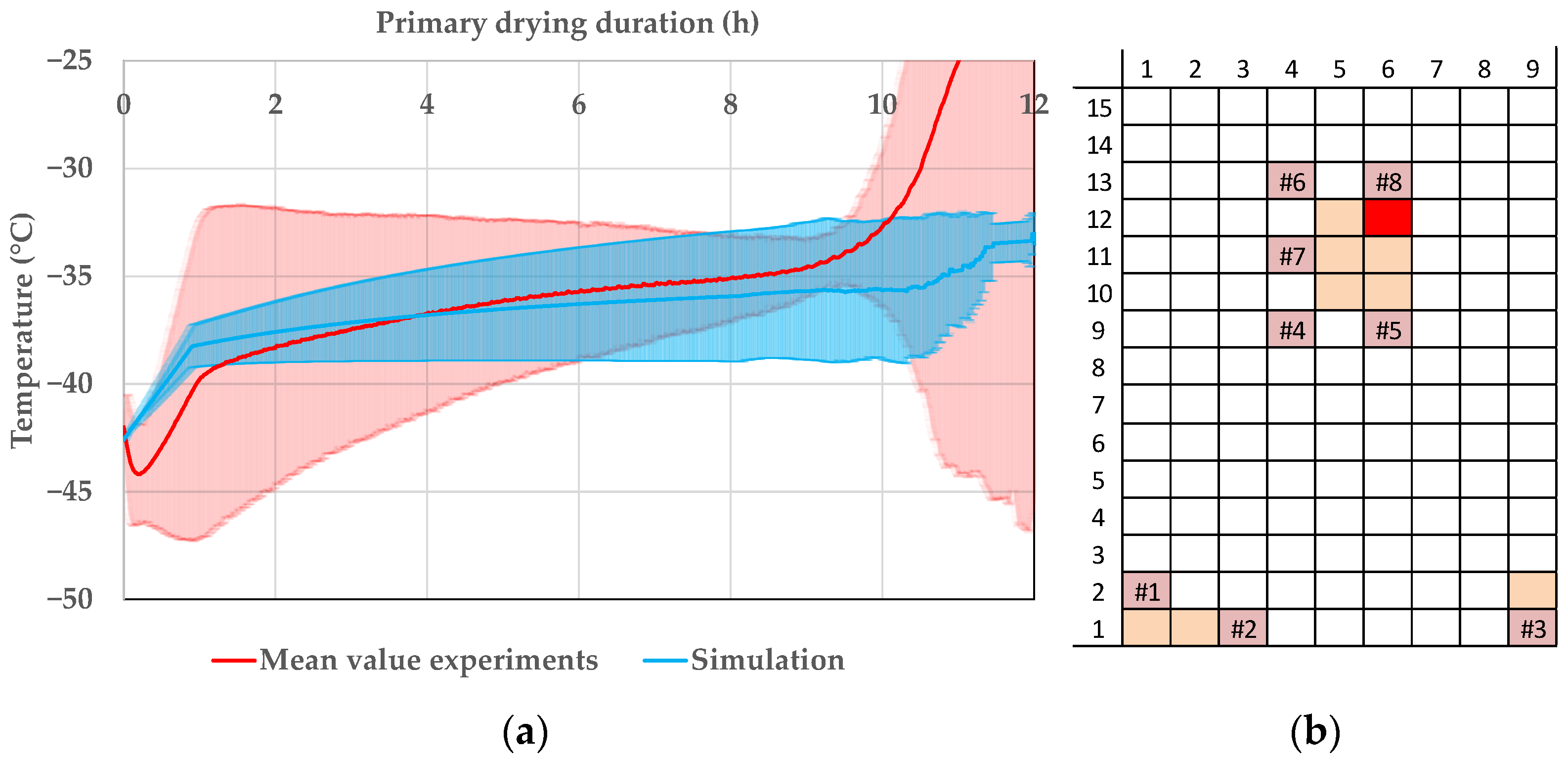
Pharmaceutics, Free Full-Text

Recommended Best Practices for Lyophilization Validation-2021 Part

Gregory SACHA, Senior Research Scientist

Gregory SACHA, Senior Research Scientist
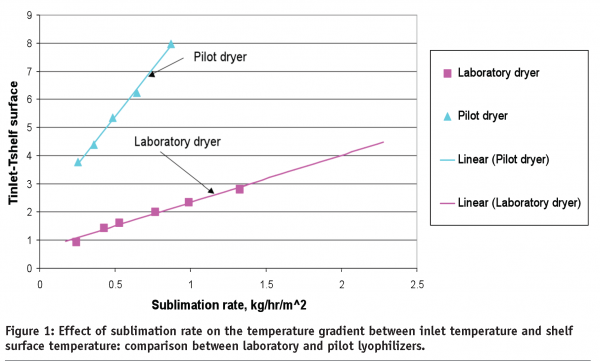
Lyophilization: cycle robustness and process tolerances, transfer and scale up - European Pharmaceutical Review









