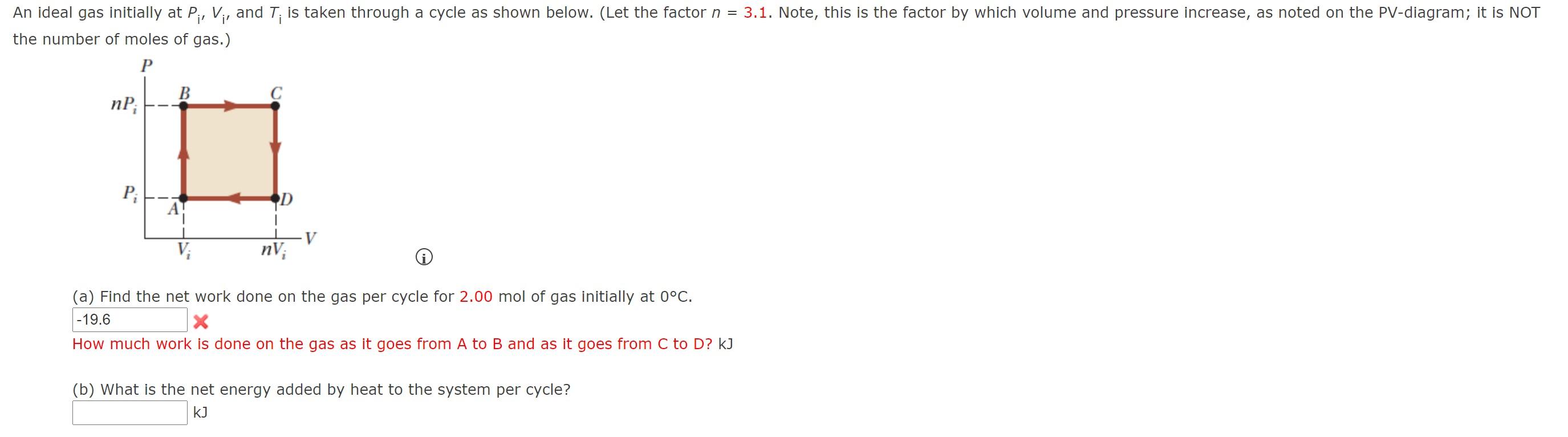
Solved An ideal gas initially at Pi, Vi, and T; is taken

An ideal gas initially P_i ,V_i , and T_i is taken through a cycle
Gas Definition, State of Matter, Properties, Structure, & Facts

SOLVED: An ideal gas initially at Pi, Vi, and Ti is taken through
Quantum thermodynamics of single particle systems
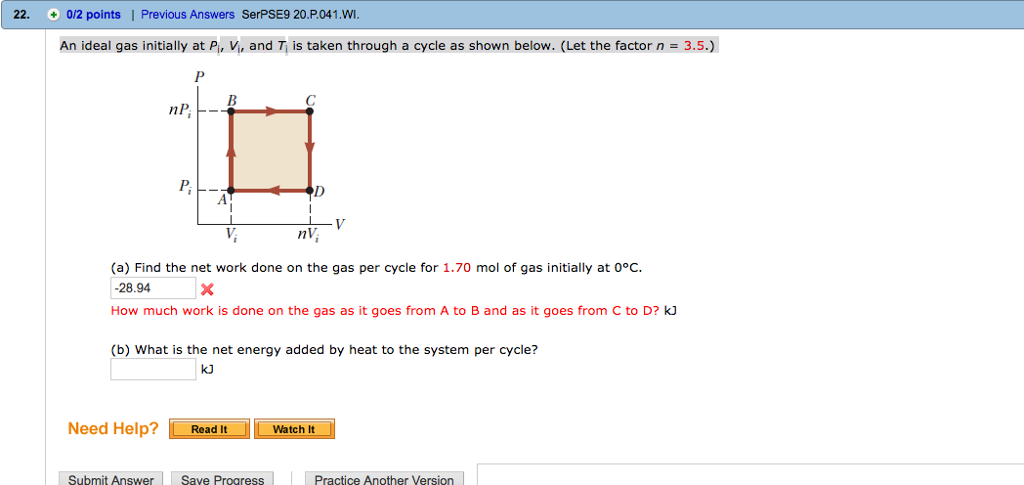
Solved An ideal gas initially at Pi, Vi, and Ti is taken
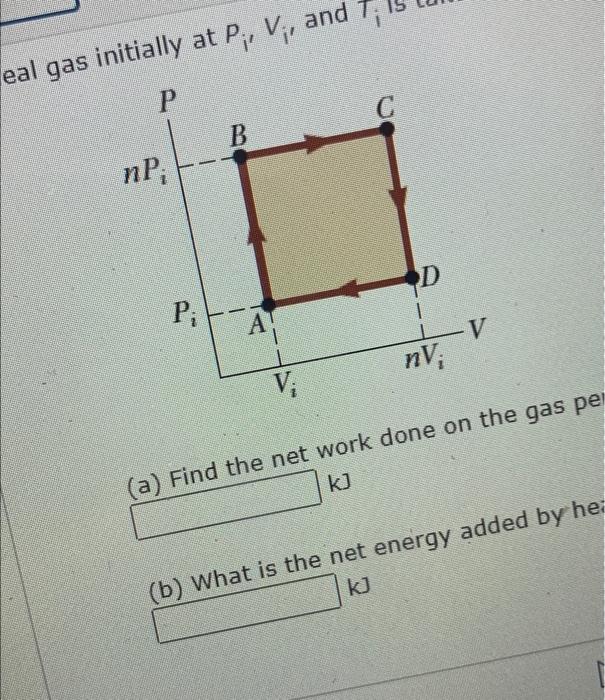
Solved An ideal gas initially at Pi, Vi, and Ti is taken

1 mole of an ideal gas undergoes reversible isothermal expansion
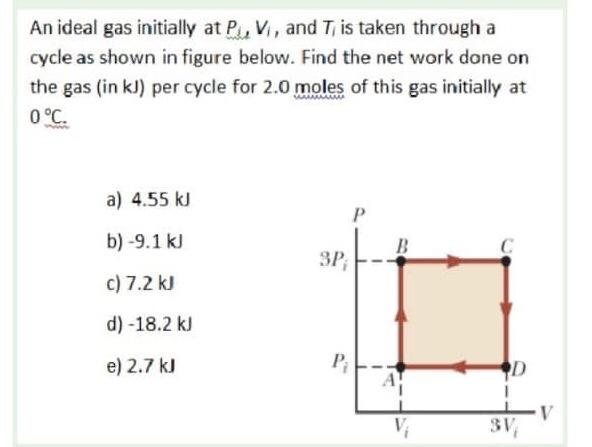
Solved An ideal gas initially at PJ, V, and Ti is taken

Relating Pressure, Volume, Amount, and Temperature: The Ideal Gas
Two moles of an ideal gas is compressed isothermally and

SOLVED: An ideal gas initially at Pi, Vi, and Ti is taken through

What is the Maxwell-Boltzmann distribution? (article)
Let's Derive the Ideal Gas Law from Scratch!








