
the compression factor one mole of a vander waals gas 0 C and 100
Click here:point_up_2:to get an answer to your question :writing_hand:the compression factor for one mole of a vander waals gas at 0 c and
Click here👆to get an answer to your question ✍️ The compression factor one mole of a vander waals gas 0 C and 100 atm pressure is found to be 0-5

Physical Chemistry The Compression Factor (Z) [w/1 example]

18. The compressibility factor one mole of a vanderwaal's gas 0°C and 100 atm pressure is found to be 0.5. Assume that the volume of gas molecule is negligible calculate the vanderwaals

2 mol of ammonia occupied a volume of 5 L at 27∘ C. Calculate the pressure if the gas obeyed van der Waals equation. a = .4.17 atm L 2 mol 2

⏩SOLVED:The compression factor (compressibility factor) for one mole…

For one mole of a van der Waals' gas when `b=0` and `T=300K`, the `pV vs 1//V` plot is shown below.
How can we calculate critical temperature, volume and pressure in terms of a and b? - Quora

At high pressure, the compressibility factor for one mole of van der w

What is the compressibility factor (Z) for 0.02 mole of a van der Waal

123. The compra compressibility factor one mole of a der Waals gas 0º C and 100 atm pressure found to be 0.5 Assume that the volume of es molecule is negligible. The

The compression factor (compressibility factor) for one mole of a Van der..

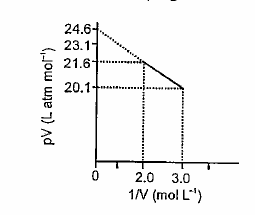
Bengali] For one mole of a van der Waals' gas when b = 0and T =300 K
The compression factor (compressibility factor) for one mole of a van der Waals' gas - Sarthaks eConnect

123. The compra compressibility factor one mole of a der Waals gas 0º C and 100 atm pressure found to be 0.5 Assume that the volume of es molecule is negligible. The
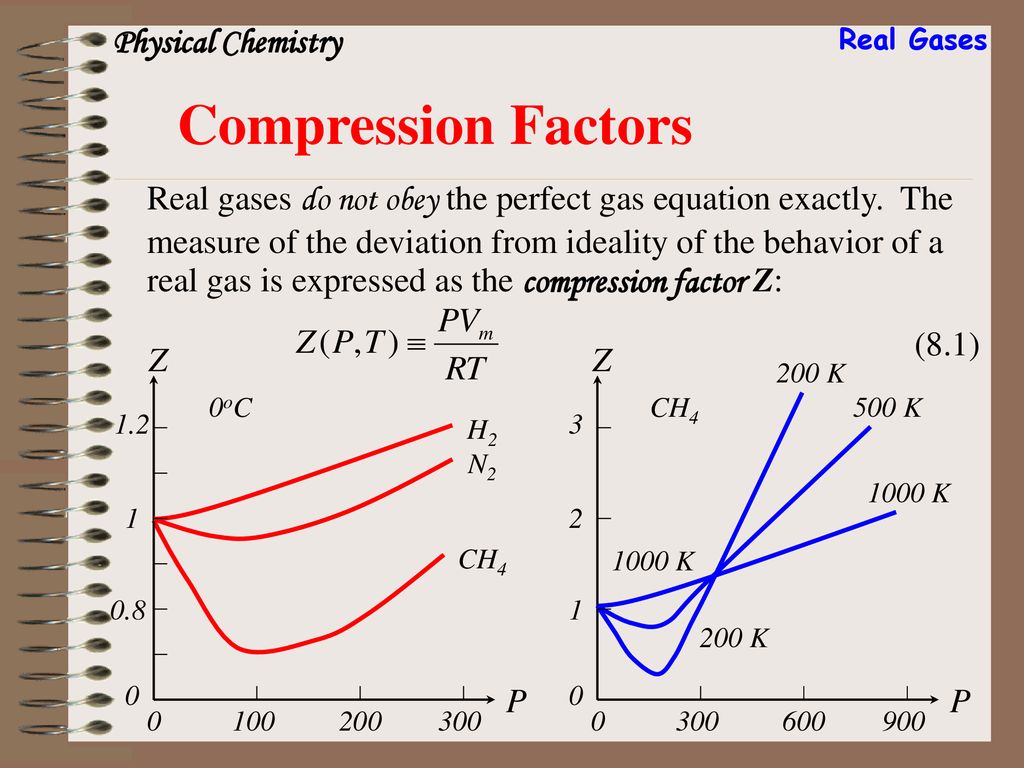






)

