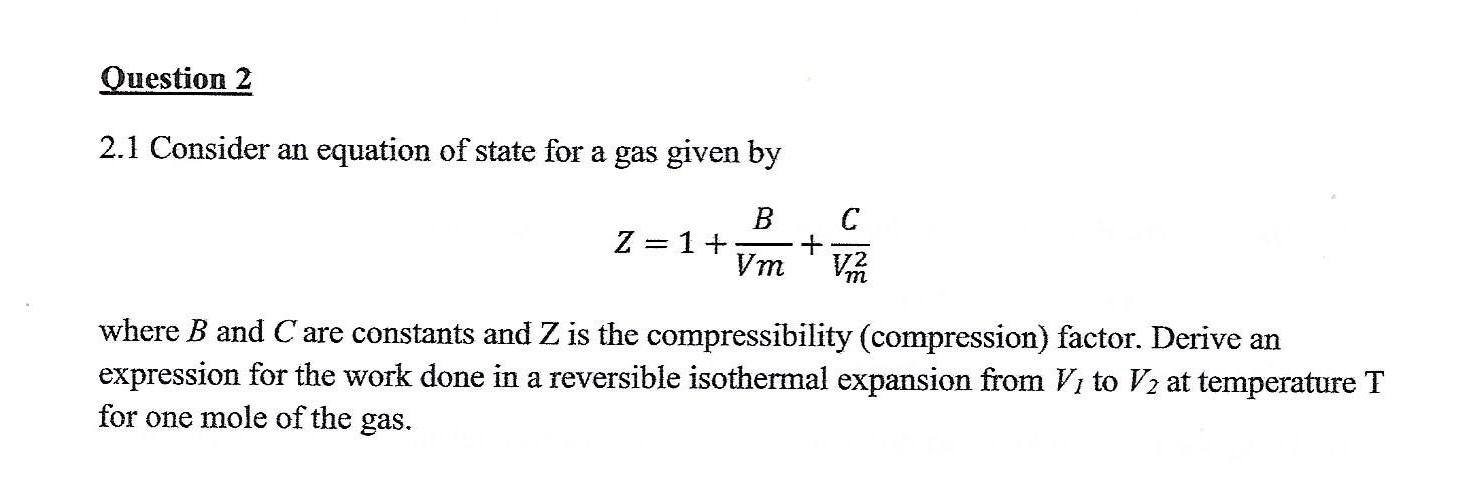If `Z` is a compressibility factor, van der Waals' equation at low
4.9
(677)
Write Review
More
$ 5.50
In stock
Description
If `Z` is a compressibility factor, van der Waals' equation at low pressure can be written as
Answer in Molecular Physics Thermodynamics for Neilmar #278440

If Z is a compressibility factor, van der Waals equation at low

Gaseous State - 2 Free MCQ Practice Test with Solutions - Chemistry

Solved The virial expansion of the compression factor (Z)

Van Der Waals Equation - an overview
The given graph represents the variation of Z (compressibility factor

If Z is compressibility factor, vander Waals equation low pressure

⏩SOLVED:Use the van der Waals equation to plot the
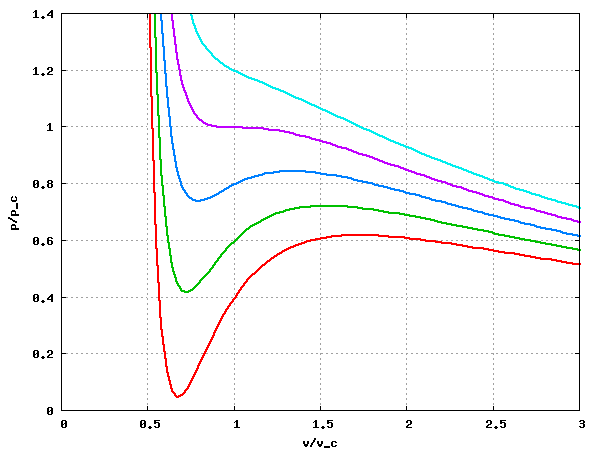
Van der Waals equation of state page on SklogWiki - a wiki for statistical mechanics and thermodynamics

Non-ideal behavior of gases (article)
Related products
You may also like