
What is the compressibility factor (Z) for 0.02 mole of a van der Waal
(d) (0.1+(1000xx(0.02)^(2))/(V^(2)))V=20xx0.02 =0.1V^(2)-0.4V+0.4=0 =V^(2)-4V+4=0 implies" "V=2L Z=(PV)/(nRT)=(0.1xx2)/(20xx0.02)=0.5
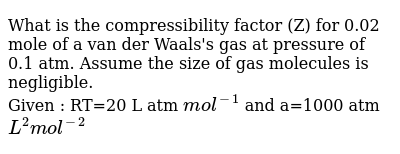
What is the compressibility factor (Z) for 0.02 mole of a van der Waal

Ratio of the rate of effusion of oxygen gas 1.5 atm to that of helium gas 4.5 atm will be (1) 1:62 (2) 1:12 Yon4 13 (3) 1:22 (4) 1:3 Compressibility factor under critical state of a gas is

PDF) Understanding on thermodynamic properties of van der Waals

Non-Ideal Gas Behavior - LabXchange

SOLVED: State (i) the ideal gas equation (ii) the van der Waal's

Two flask A and B of equal volumes maintained at temperature 300K an

PEDULCE UUUUUUUU 3. A 1 litre vessel contains 2 moles of a vanderwaal's gas. Given data : a = 2.5 atm-Lmole - T= 240 K b = 0.4 L-mole- RT = 20

THE 3rd STATE OF MATTER – What is a Real Gas? – Computer Aided

PDF) Understanding on thermodynamic properties of van der Waals

If Z is a compressibility factor, van der Waals equation at low

jo 22] What is the compressibility factor (Z) 0.02 mole of a van der Waals' gas pressure of 0.1 atm. Assume the size of gas molecules is negligible.

Illallis salle ( D uallyes upi culcauiy foecleases B-16. What is the compressibility factor (Z) 0.02 mole of a van der Waals' gas pressure of 04 atm. Assume the size of gas









