
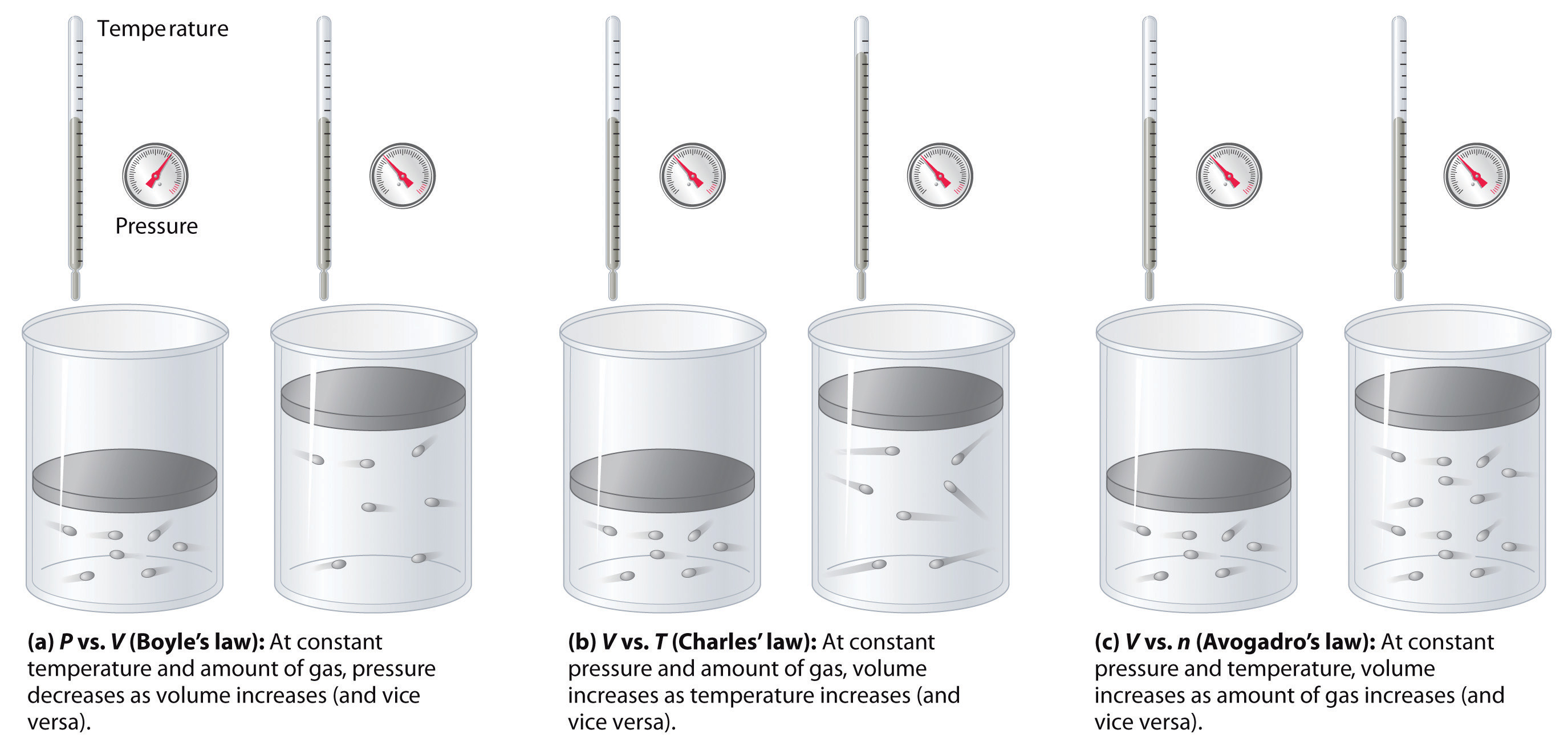
Two closed bulbs of equal volume V containing an ideal gas initially at pressure P i and temperature T 1 are connected through a narrow tube of negligible volume as shown in
Two closed bulbs of equal volume V containing an ideal gas initially at pressure P i and temperature T 1 are connected through a narrow tube of negligible volume as shown in the figure below. The temperature of one of the bulbs is then raised to T 2. The final pressure Pf is :P i T 1 T 2/ T 1+ T 2B. 2 P i T 1/ T 1+ T 2C. 2 P i T 1 T 2/ T 1+ T 2D. 2 P i T 2/ T 1+ T 2
Two closed bulbs of equal volume V containing an ideal gas initially at pressure P i and temperature T 1 are connected through a narrow tube of negligible volume as shown in the figure below- The temperature of one of the bulbs is then raised to T 2- The final pressure Pf is -P i T 1 T 2- T 1- T 2B- 2 P i T 1- T 1- T 2C- 2 P i T 1 T 2- T 1- T 2D- 2 P i T 2- T 1- T 2
The correct option is D 2P_i ( T_2T_1+T_2 )Since the above system is a closed one, the total number of moles of the ideal gas will be equal before and after th

Two closed bulbs of equal volume V containing an ideal gas initially at pressure P i and temperature T 1 are connected through a narrow tube of negligible volume as shown in

Two closed bulbs of equal volume (V) containing an ideal gas initia
Two closed bulbs of equal volume (V) containing an ideal gas initially at pressure pi and temperature T1 are connected - Sarthaks eConnect

Answered: The pressure, volume, and temperature…
Gases

A closed vessel A having volume V contains N(2) at pressure P and temp
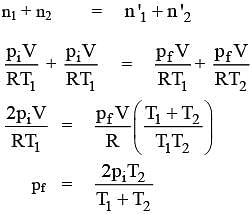
Two closed bulbs of equal volume (V) containing an ideal gas initially at pressure pi and temperature T1 are connected through a narrow tube of negligible volume as shown in the figure
Telugu] Two closed vessel A and B of equal volume containing air at p

Thermodynamics: An Engineering Approach - 5th Edition - Part II by 黑傑克 - Issuu
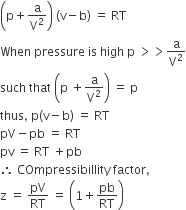
The compressibility factor for a real gas at high pressure is f

Two closed bulbs of equal volume V containing an ideal gas

Gaseous State - 2 Free MCQ Practice Test with Solutions - Chemistry
Bengali] Two bulbs of equal volume are connected by a narrow tube of









