
An ideal gas initially P_i ,V_i , and T_i is taken through a cycle
Click here:point_up_2:to get an answer to your question :writing_hand:an ideal gas initially at pi vi and ti is taken through a cycle
Click here👆to get an answer to your question ✍️ An ideal gas initially P-i -V-i - and T-i is taken through a cycle as shown in Figure- -a- Find the net work done on the gas per cycle 1-00 mol of gas initially 0-0C- -b- What is the net energy added by heat to the gas per cycle

A 1.00 mol sample of monoatomic ideal gas is take through the cycle shown. At point A, the pressure, volume and temperature are P_i, V_i and T_i respectively. In terms of R


The First Law and Enthalpy

Process integration, energy and exergy analyses of a novel integrated system for cogeneration of liquid ammonia and power using liquefied natural gas regasification, CO2 capture unit and solar dish collectors - ScienceDirect

Nanoreactors in action for a durable microactuator using spontaneous combustion of gases in nanobubbles
1st law
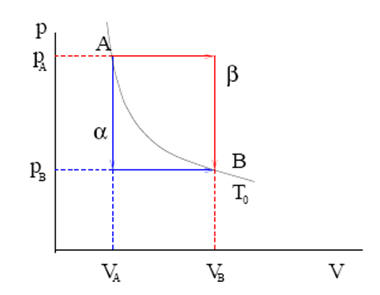
1st law

⏩SOLVED:An ideal gas initially at Pi, Vi, and Ti is taken through a…
Solving a Monatomic Ideal Gas Expansion Problem

Progress in interfacial solar steam generation using low-dimensional and biomass-derived materials - ScienceDirect








