Solved 14. The latent heat of vaporization of isopropyl
Answer to Solved 14. The latent heat of vaporization of isopropyl

Regain flood adaptation in rice through a 14-3-3 protein OsGF14h

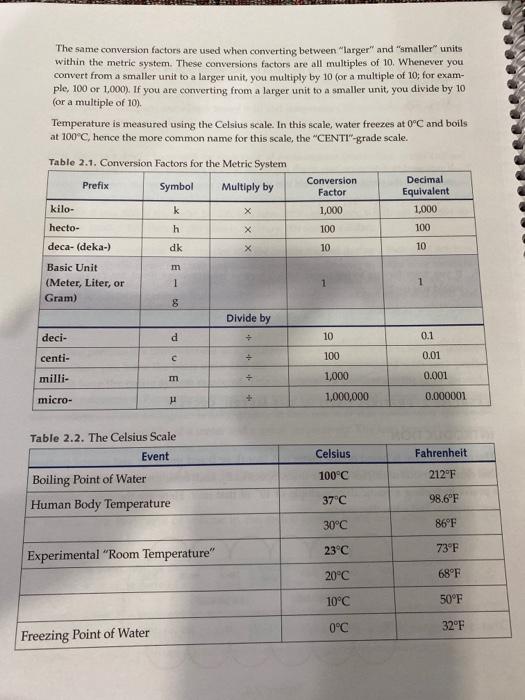
Estimation of Heat of Vaporization

Heat of Vaporization of Water vs. Rubbing Alcohol

SOLVED: Determine the total amount of heat released (in kJ) when 485 g of isopropyl alcohol (C3H8O) are cooled from the gas state at 100 °C to liquid at -42.0 °C. The
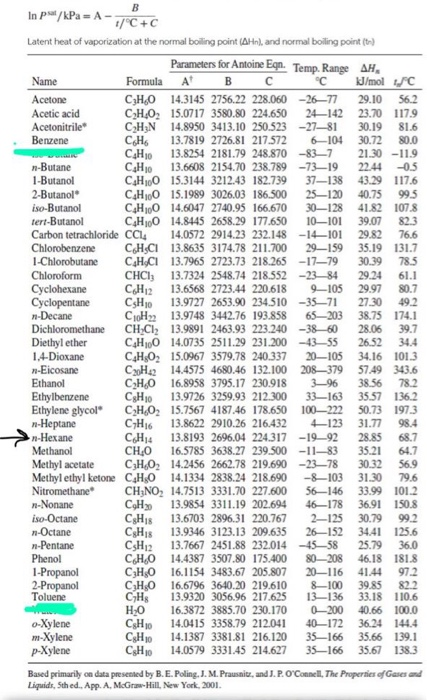
Solved Binary system 1-chlorobutane/chlorobenzene conforms
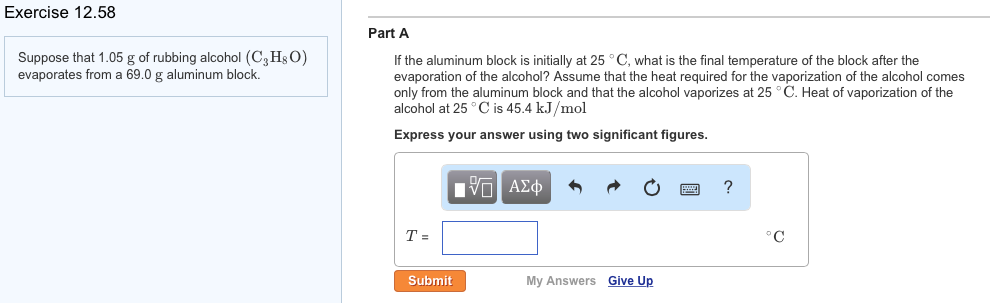
Solved Suppose that 1.05 g of rubbing alcohol (C3H8O)
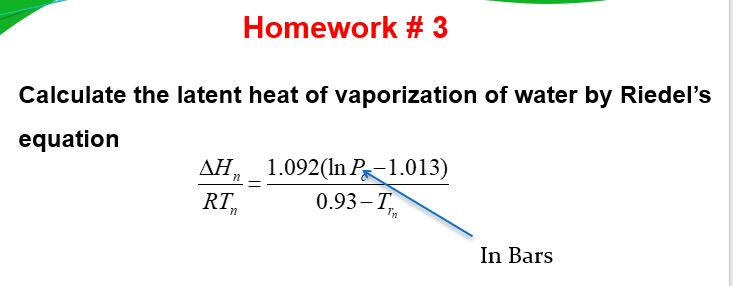
Solved Homework # 3 Calculate the latent heat of
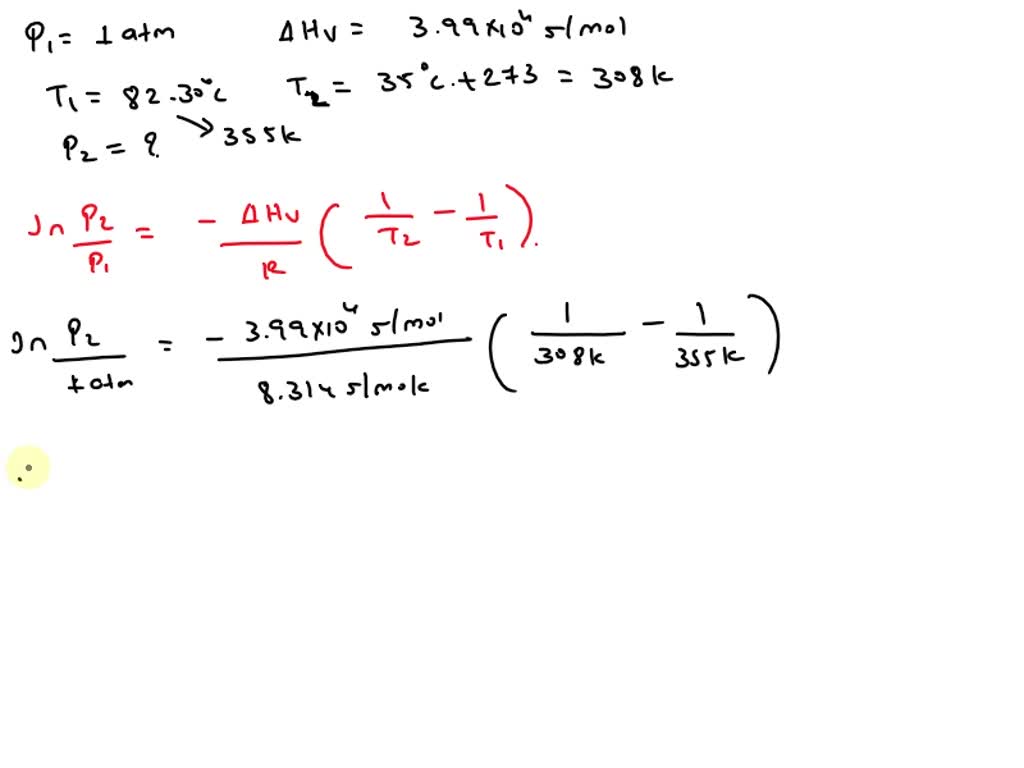
SOLVED: Isopropyl alcohol has a heat of vaporization of 3.99 * 10^3 mol^-1 and a boiling point of 82.30 °C at 1.000 atm. Using the Clausius-Clapeyron equation, calculate the vapor pressure of
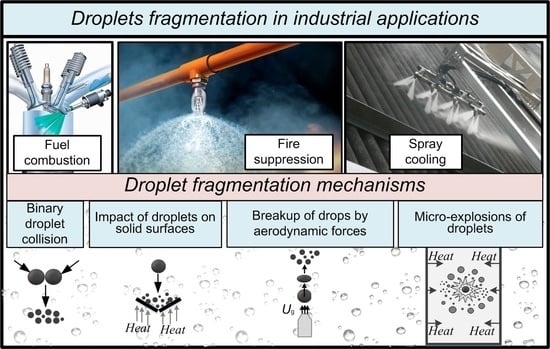
Energies, Free Full-Text

Solved Suppose that 1.02 g of rubbing alcohol (C3H8O)

Isopropyl Alcohol, (CH3)2CHOH

Clean production of isopropyl myristate: A cutting-edge enzymatic approach with a holistic techno-economic evaluation - ScienceDirect

Solved 1. (25 pts) The latent eats of vaporization in J/g

Materials Proceedings, Free Full-Text

SOLVED: Part 3: Latent Heat of Vaporization of Water Data: Mass of water initially: 5.8 g Mass of aluminum cup: 543.3 g Initial water-Al temperature: Twi Mass of water vapor added: 26.8