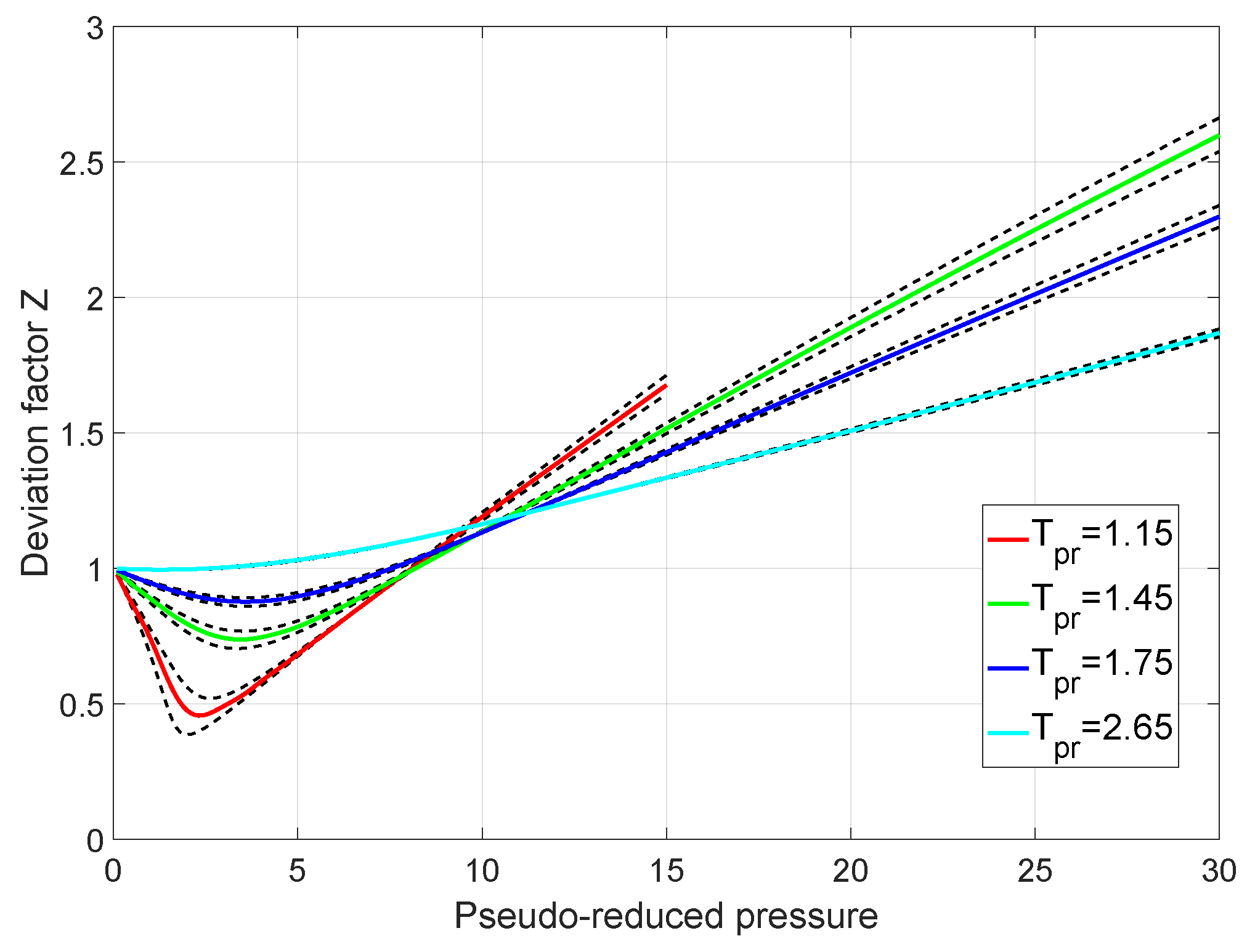
What is the value of compressibility factor in terms of vander
What is the value of compressibility factor in terms of vander waal cons†an t at different conditions of pressure and volume?Why is Z>1 for H2 and He gas
What is the value of compressibility factor in terms of vander waal cons-an t at different conditions of pressure and volume-Why is Z-1 for H2 and He gas
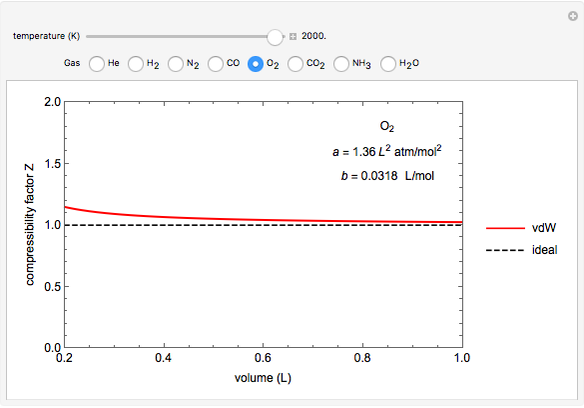
Compressibility Factors for van der Waals Gases - Wolfram Demonstrations Project

Compressibility Factors for van der Waals Gases - Wolfram Demonstrations Project

Gas compressibility factor Z: Ideal gas vs Real gas
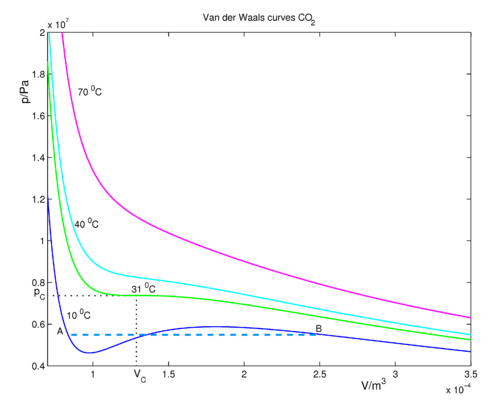
Van der Waals equation - Knowino

If `Z` is a compressibility factor, van der Waals' equation at low pressure can be written as

Non-Ideal Gas Behavior Chemistry: Atoms First
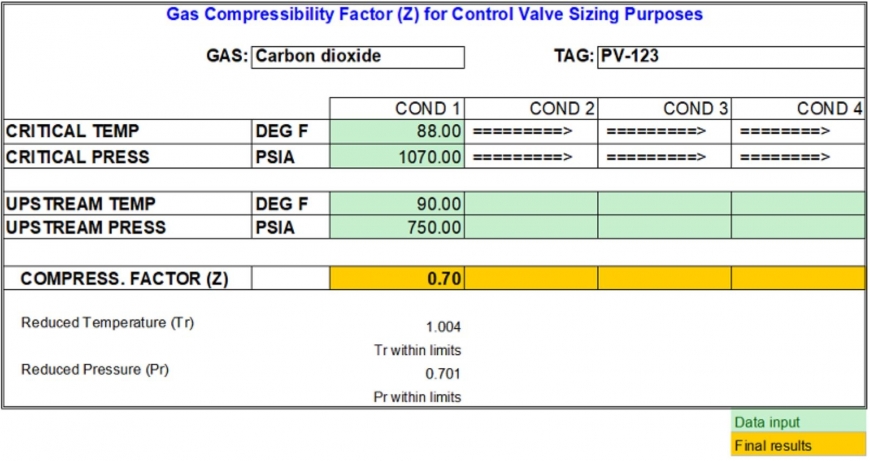
Gas Compressibility Factor and Control Valve Sizing
What is the value of compressibility factor in terms of vander waal cons†an t at different conditions of pressure and volume?Why is Z>1 for H2 and He gas

Deviations from ideal gas behaviour, intermolecular forces, Van der Waals equation of state, compressibility factors and the critical pressure and critical temperature of a gas revision notes doc brown's chemistry UK advanced
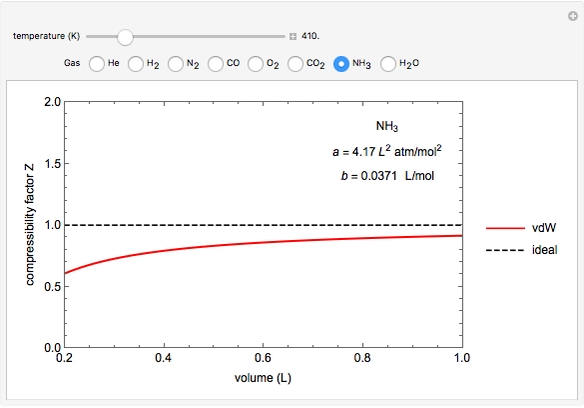
Compressibility Factors for van der Waals Gases - Wolfram Demonstrations Project
Compressibility Factors for van der Waals Gases - Wolfram Demonstrations Project

The van der Waals equation (video)

At very high pressure, the compressibility factor of 1 mole of a

plotting - How to plot Compressibility factor Z vs Pressure P using ParametricPlot? - Mathematica Stack Exchange